


Introduction
Spectroscopy is the scientific study of the interaction between electromagnetic radiation and matter. It is one of the most important techniques in physics, chemistry, astronomy, and materials science. Spectroscopy allows scientists to analyze the composition, structure, and properties of substances by examining how they absorb, emit, or scatter light.
The term spectroscopy originates from the Latin word spectrum, meaning appearance or image, and the Greek word skopein, meaning to observe. Essentially, spectroscopy involves studying how light behaves when it interacts with atoms, molecules, or solids.
Spectroscopy has played a fundamental role in many scientific discoveries. Early work by scientists such as Joseph von Fraunhofer revealed dark lines in the solar spectrum, known as Fraunhofer lines, which provided clues about the chemical composition of the Sun.
Later developments in quantum mechanics helped explain the origin of spectral lines through the work of scientists like Niels Bohr.
Today, spectroscopy is widely used in fields such as astronomy, environmental science, medical diagnostics, and industrial analysis.
Basic Principles of Spectroscopy
Spectroscopy is based on the interaction between electromagnetic radiation and matter.
When atoms or molecules interact with radiation, several processes can occur:
- Absorption of energy
- Emission of radiation
- Scattering of light
These processes produce characteristic spectral patterns that reveal information about the substance being studied.
Each element has a unique set of energy levels. When electrons transition between these energy levels, specific wavelengths of radiation are absorbed or emitted.
The wavelength or frequency of radiation involved in the transition is related to the energy difference between the levels.
Electromagnetic Radiation
Spectroscopy primarily deals with electromagnetic radiation, which includes a wide range of wavelengths and frequencies.




Electromagnetic radiation includes:
- Radio waves
- Microwaves
- Infrared radiation
- Visible light
- Ultraviolet radiation
- X-rays
- Gamma rays
Each region of the spectrum is used for different spectroscopic techniques.
The relationship between wavelength, frequency, and wave speed is given by:
genui{“math_block_widget_common_keywords”:{“content”:”v = f\lambda”}}
This relationship shows that wavelength and frequency are inversely related.
Types of Spectra
Spectroscopy produces several types of spectra depending on the interaction between radiation and matter.
Emission Spectrum




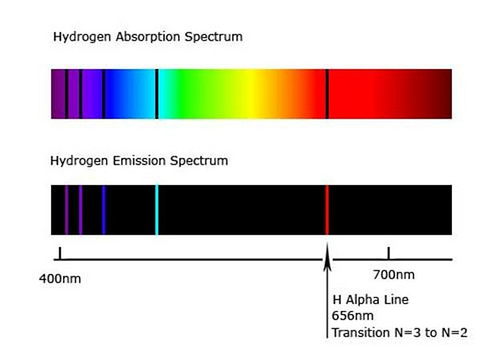
An emission spectrum is produced when atoms or molecules emit radiation after being excited.
In this process:
- Electrons absorb energy.
- Electrons move to higher energy levels.
- When electrons return to lower energy levels, photons are emitted.
The emitted wavelengths form bright lines on a dark background.
Each element produces a unique emission spectrum.
Absorption Spectrum




An absorption spectrum occurs when atoms absorb specific wavelengths from a continuous spectrum.
This produces dark lines in the spectrum where light has been absorbed.
Absorption spectra are important for identifying elements in stars and gases.
Continuous Spectrum
A continuous spectrum contains all wavelengths of light within a range.
This type of spectrum is produced by hot, dense objects such as incandescent solids or liquids.
The spectrum appears as a smooth band of colors without gaps.
Atomic Spectroscopy
Atomic spectroscopy studies the interaction of radiation with individual atoms.
Electrons in atoms occupy discrete energy levels.
When electrons transition between these levels, photons of specific energies are emitted or absorbed.
The energy of a photon is given by:
[
E = hf
]
where:
- (E) = energy
- (h) = Planck’s constant
- (f) = frequency
These transitions produce characteristic spectral lines unique to each element.
Molecular Spectroscopy

Molecular spectroscopy studies the interaction of radiation with molecules.
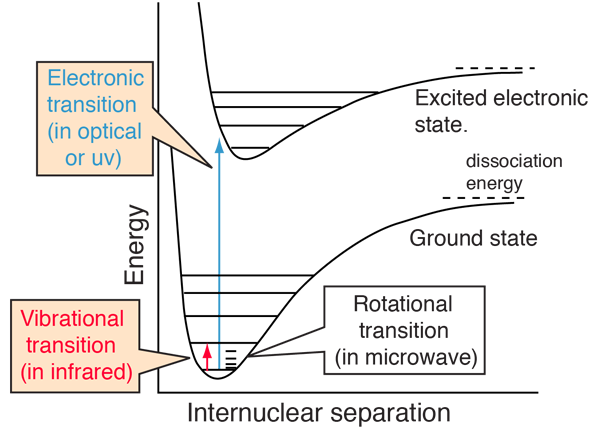
Molecules have more complex energy structures than atoms.
Molecular energy transitions include:
- Rotational transitions
- Vibrational transitions
- Electronic transitions
These transitions occur at different regions of the electromagnetic spectrum.
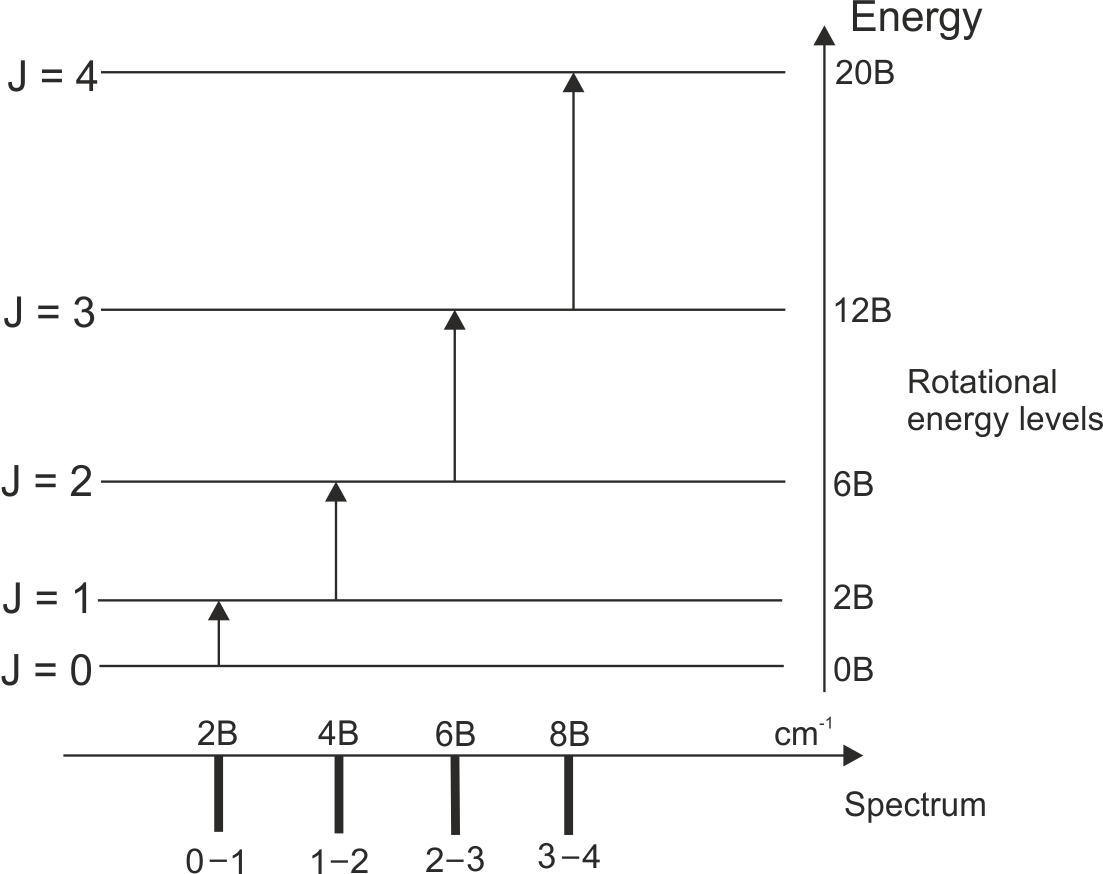
Rotational Spectroscopy
Rotational spectroscopy involves transitions between rotational energy levels of molecules.
This type of spectroscopy typically occurs in the microwave region of the electromagnetic spectrum.
It provides information about molecular structure and bond lengths.
Vibrational Spectroscopy
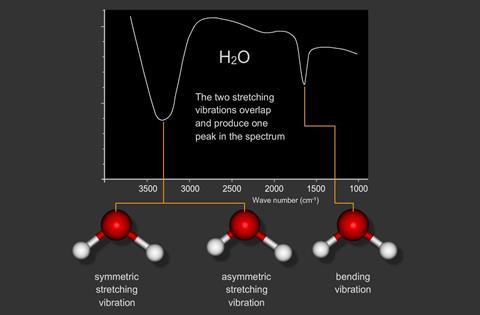
Vibrational spectroscopy studies vibrations of atoms within molecules.
These transitions occur in the infrared region.
Infrared spectroscopy is widely used to identify chemical compounds.
Electronic Spectroscopy
Electronic spectroscopy involves transitions between electronic energy levels in atoms and molecules.
These transitions occur in the visible and ultraviolet regions.
This type of spectroscopy is commonly used in chemical analysis and materials research.
Instrumentation in Spectroscopy



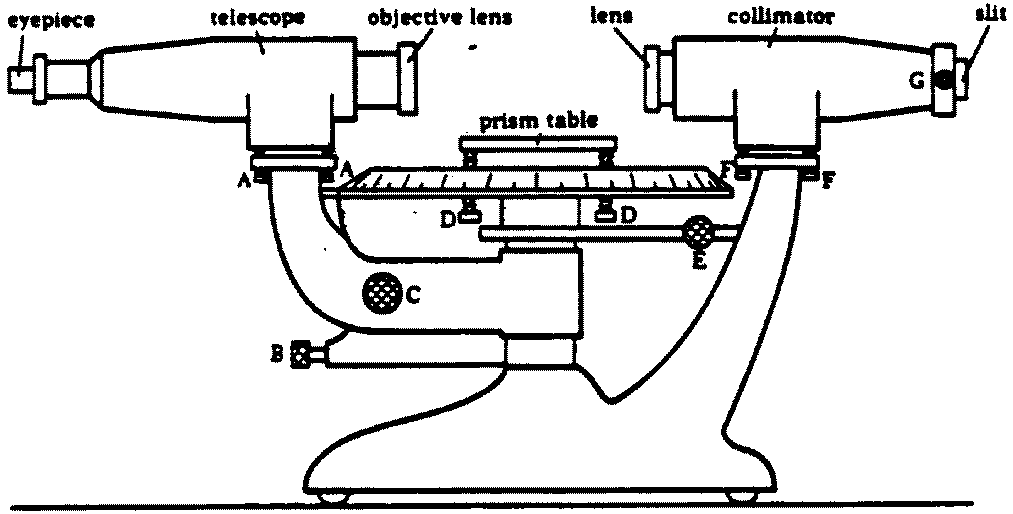
Spectroscopic measurements are performed using instruments called spectrometers.
A typical spectrometer contains several key components:
- Radiation source
- Sample holder
- Wavelength selector (prism or diffraction grating)
- Detector
- Data recording system
The spectrometer separates light into its component wavelengths and measures their intensity.
Spectroscopic Techniques
Several spectroscopic techniques are widely used in science and technology.
Infrared Spectroscopy
Used to study molecular vibrations and identify functional groups in molecules.
Ultraviolet–Visible Spectroscopy
Used to study electronic transitions in atoms and molecules.
Nuclear Magnetic Resonance Spectroscopy
Used to determine molecular structure.
Raman Spectroscopy
Based on scattering of light by molecules.
Provides information about vibrational modes.
Mass Spectrometry
Although not strictly optical spectroscopy, it analyzes molecules based on mass-to-charge ratio.
Applications of Spectroscopy
Spectroscopy has numerous applications across scientific disciplines.
Astronomy
Spectroscopy is used to determine the composition of stars and galaxies.
By analyzing spectral lines, astronomers can identify elements present in celestial objects.
Chemistry
Spectroscopy helps identify chemical compounds and analyze molecular structures.
Environmental Science
Spectroscopic techniques detect pollutants and monitor atmospheric gases.
Medicine
Spectroscopy is used in medical imaging and diagnostic techniques.
Examples include magnetic resonance imaging and laser spectroscopy.
Materials Science
Spectroscopy helps study electronic properties and structures of materials.
Forensic Science
Spectroscopic analysis can identify substances in criminal investigations.
Spectroscopy and Quantum Mechanics
The development of spectroscopy played a key role in the development of quantum mechanics.
Spectral lines could only be explained by quantized energy levels.
Quantum theory explains how electrons transition between energy states and produce spectral lines.
Modern Advances in Spectroscopy
Modern spectroscopy uses advanced technologies such as:
- Laser spectroscopy
- Fourier transform spectroscopy
- High-resolution spectrometers
- Space-based spectroscopic instruments
These techniques provide extremely precise measurements of atomic and molecular properties.
Spectroscopy in Astrophysics
Spectroscopy allows astronomers to determine:
- Chemical composition of stars
- Temperature of stellar atmospheres
- Motion of galaxies through Doppler shifts
This technique has revealed the presence of many elements throughout the universe.
Conclusion
Spectroscopy is a powerful scientific technique used to study the interaction between electromagnetic radiation and matter. By analyzing how atoms and molecules absorb, emit, or scatter light, scientists can determine the composition, structure, and physical properties of substances.
Different types of spectroscopy—including atomic, molecular, infrared, ultraviolet, and Raman spectroscopy—provide valuable insights across many scientific fields. Spectroscopic methods have been essential in the development of modern physics and chemistry and continue to play a critical role in research and technological advancement.
As scientific instrumentation and computational methods continue to advance, spectroscopy remains one of the most important tools for exploring the microscopic world and understanding the fundamental behavior of matter.
