Introduction
The partition function is one of the most important concepts in statistical mechanics. It acts as a bridge between the microscopic properties of a physical system and its macroscopic thermodynamic behavior. Through the partition function, it becomes possible to derive quantities such as internal energy, entropy, pressure, heat capacity, and free energy directly from the statistical properties of particles.
Statistical mechanics studies systems composed of a very large number of microscopic particles such as atoms or molecules. These particles can occupy different energy states depending on their physical interactions and environmental conditions such as temperature and volume. The partition function provides a mathematical way to account for all possible energy states and determine how likely each state is.
In simple terms, the partition function summarizes all the possible microscopic configurations of a system and their associated energies. Once the partition function is known, almost every thermodynamic property of the system can be calculated.
Partition functions are used extensively in many areas of physics and chemistry, including:
- Statistical thermodynamics
- Quantum mechanics
- Molecular physics
- Chemical thermodynamics
- Solid-state physics
They are particularly useful in systems where many microscopic configurations contribute to the overall behavior of the system.
Concept of Energy States in Statistical Mechanics



In statistical mechanics, a system can exist in many different microstates, each corresponding to a specific energy value. These energy levels represent the possible configurations of particles within the system.
For example:
- Molecules in a gas can move with different kinetic energies.
- Atoms in a solid can vibrate with different amplitudes.
- Electrons in atoms can occupy different quantum energy levels.
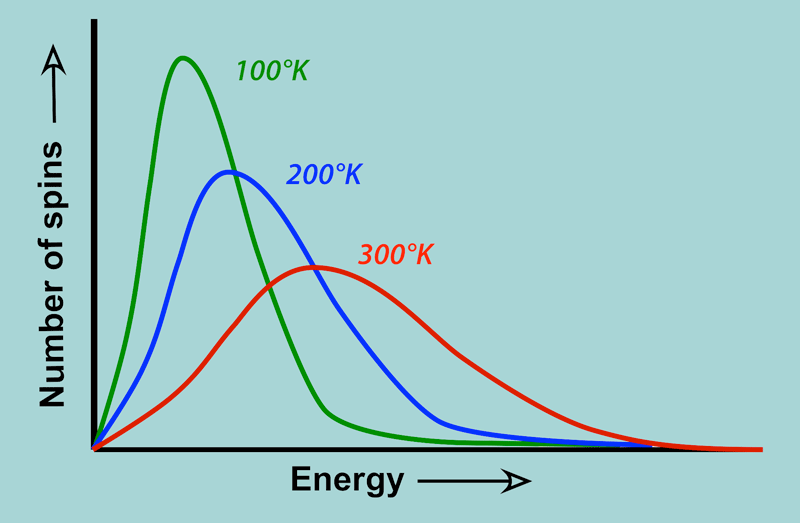
The probability that a system occupies a particular energy state depends on the temperature and the energy of that state.
At thermal equilibrium, the probability distribution of energy states follows the Boltzmann distribution.
Boltzmann Distribution


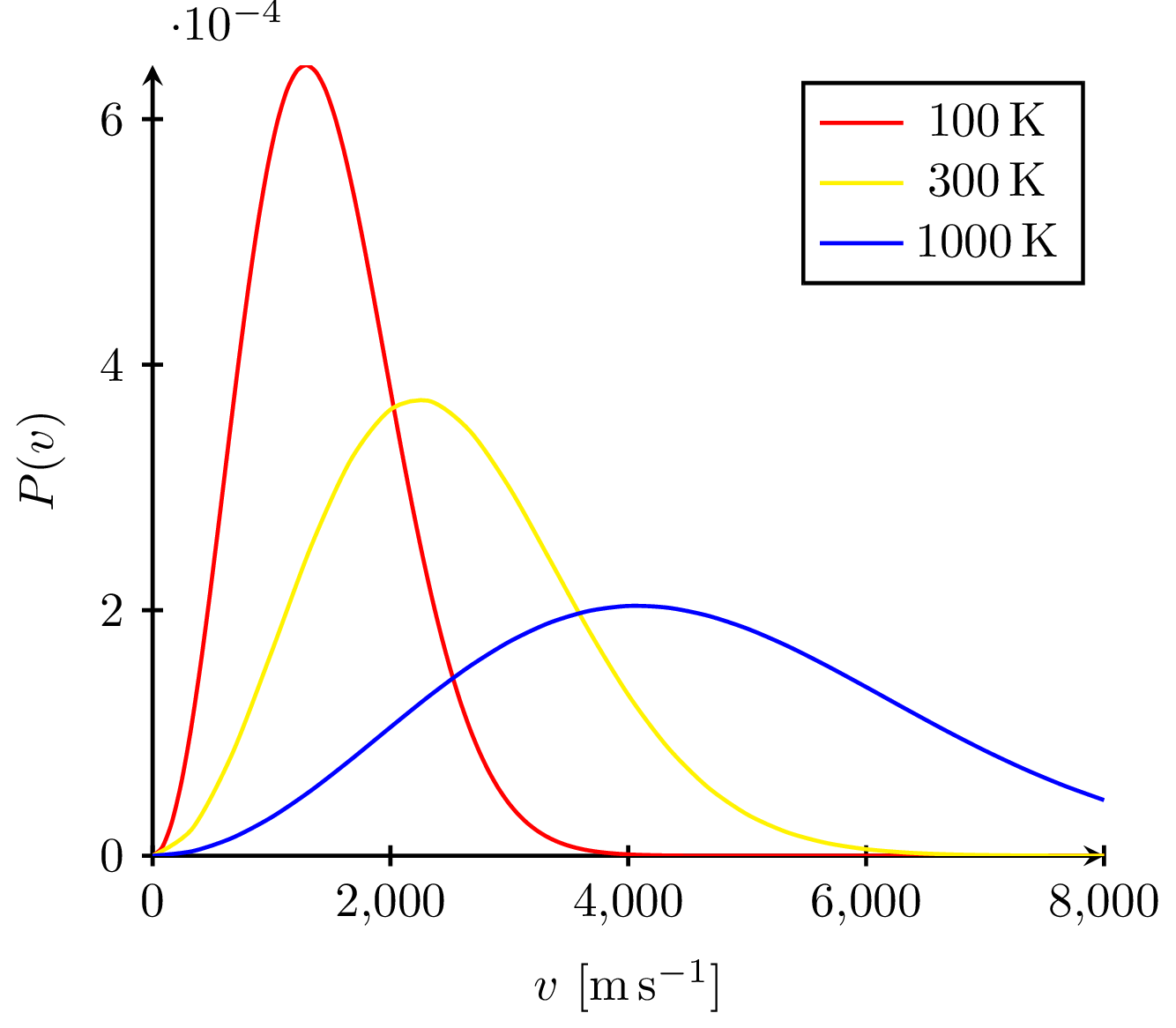
The Boltzmann distribution describes the probability that a system occupies a state with energy (E_i).
[
P_i = \frac{e^{-E_i / kT}}{Z}
]
Where:
- (P_i) = probability of state (i)
- (E_i) = energy of the state
- (k) = Boltzmann constant
- (T) = temperature
- (Z) = partition function
The exponential factor (e^{-E_i/kT}) is called the Boltzmann factor.
States with lower energy are more probable, while higher-energy states become more likely as temperature increases.
Definition of the Partition Function
The partition function is defined as the sum of Boltzmann factors for all possible states.
[
Z = \sum_i e^{-E_i / kT}
]
Where:
- (Z) = partition function
- (E_i) = energy of state (i)
The partition function serves as a normalization constant that ensures the probabilities of all states sum to one.
Because it contains information about every possible energy state of the system, the partition function encodes the entire thermodynamic behavior of the system.
Physical Meaning of the Partition Function




The partition function represents a weighted sum of all possible microscopic configurations of a system.
Each configuration contributes to the partition function according to its Boltzmann factor.
Important interpretations include:
- It measures the number of accessible states at a given temperature.
- It determines the probability of different energy states.
- It links microscopic physics to macroscopic thermodynamic quantities.
As temperature increases, higher energy states contribute more significantly to the partition function.
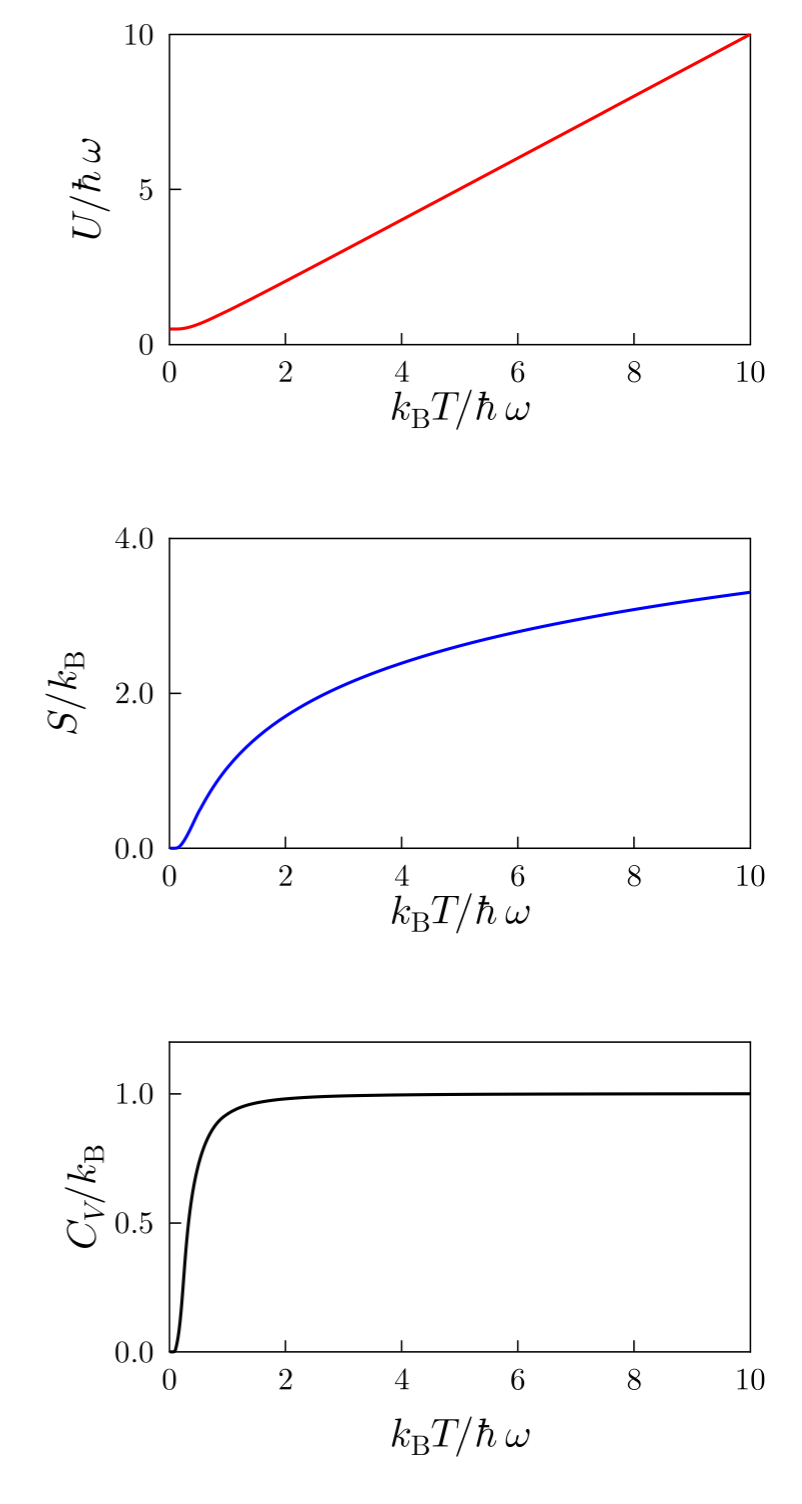
Thermodynamic Quantities from the Partition Function




One of the most powerful features of the partition function is that it allows calculation of many thermodynamic quantities.
Internal Energy
[
U = – \frac{\partial}{\partial \beta} \ln Z
]
Where ( \beta = 1/kT ).
Helmholtz Free Energy
[
F = -kT \ln Z
]
Entropy
[
S = – \left(\frac{\partial F}{\partial T}\right)_V
]
Heat Capacity
[
C = \frac{\partial U}{\partial T}
]
These equations demonstrate that once the partition function is known, all thermodynamic properties can be derived.
Types of Partition Functions




In molecular systems, the total partition function can be separated into contributions from different types of motion.
Translational Partition Function
Describes motion of particles through space.
Rotational Partition Function
Describes rotational motion of molecules.
Vibrational Partition Function
Describes molecular vibrations.
Electronic Partition Function
Describes electronic energy levels.
The total partition function is the product of these contributions.
Classical and Quantum Partition Functions



Partition functions can be calculated using either classical or quantum mechanics.
Classical Partition Function
Used when energy levels form a continuous spectrum.
Quantum Partition Function
Used when energy levels are discrete.
Quantum partition functions are especially important for atoms, molecules, and low-temperature systems.
Partition Functions in Different Ensembles




Different statistical ensembles have different partition functions.
Canonical Ensemble
Used for systems at constant temperature, volume, and particle number.
Grand Canonical Ensemble
Used for systems where particle number can vary.
Microcanonical Ensemble
Used for isolated systems with fixed energy.
Each ensemble provides different ways to analyze thermodynamic systems.
Applications of Partition Functions

Partition functions are widely used in many scientific fields.
Chemistry
Predicting reaction equilibrium and molecular properties.
Molecular Physics
Analyzing molecular vibrations and rotations.
Solid-State Physics
Studying phonons and electron behavior.
Astrophysics
Modeling gases in stars and planetary atmospheres.
Partition functions provide a powerful tool for linking microscopic physics to observable phenomena.
Importance in Modern Physics
Partition functions are central to statistical mechanics and thermodynamics.
They provide a unified framework for understanding complex systems composed of many particles.
The concept also plays a role in advanced fields such as:
- Quantum field theory
- Condensed matter physics
- Cosmology
Many modern theoretical models rely on partition functions to describe physical systems.
Conclusion
The partition function is one of the most fundamental concepts in statistical mechanics. It provides a mathematical framework for connecting the microscopic energy states of a system with its macroscopic thermodynamic properties.
By summing over all possible energy states weighted by Boltzmann factors, the partition function captures the statistical behavior of particles in thermal equilibrium. From this single function, it is possible to derive important thermodynamic quantities such as internal energy, entropy, free energy, and heat capacity.
Partition functions are essential tools in physics, chemistry, and materials science, helping scientists understand complex systems ranging from molecular gases to quantum materials and astrophysical objects.
