Introduction to Thermodynamics
Thermodynamics is a fundamental branch of physics and chemistry that deals with the study of energy, heat, work, and their transformations. It explains how energy moves within a system and how it interacts with its surroundings. The word thermodynamics comes from the Greek words therme meaning heat and dynamis meaning power.
Thermodynamics plays a crucial role in understanding many natural and technological processes. From engines and refrigerators to biological metabolism and atmospheric processes, thermodynamic principles help explain how energy is transferred and transformed.
In chemistry, thermodynamics helps determine whether a chemical reaction will occur spontaneously and how much energy will be released or absorbed during the reaction. In physics and engineering, it is used to design engines, turbines, power plants, and refrigeration systems.
The field of thermodynamics developed during the 19th century through the work of scientists studying steam engines and heat engines. Their investigations led to the discovery of fundamental laws governing energy transfer.
Thermodynamics is primarily concerned with macroscopic properties of systems such as:
- Temperature
- Pressure
- Volume
- Internal energy
- Enthalpy
- Entropy
These quantities describe the energy state of a system and how it changes during physical or chemical processes.
1. Thermodynamic Systems
A thermodynamic system is a specific portion of the universe that is chosen for study. Everything outside the system is known as the surroundings.
For example:
- A gas inside a cylinder can be considered the system.
- The cylinder walls and external environment are the surroundings.
Understanding the interaction between a system and its surroundings is essential in thermodynamics.
Types of Thermodynamic Systems
Thermodynamic systems are classified based on the exchange of matter and energy.
Open System
An open system can exchange both energy and matter with its surroundings.
Examples:
- Boiling water in an open pot
- Human body
- Rivers and oceans
Open systems are common in biological and environmental processes.
Closed System
A closed system can exchange energy but not matter with its surroundings.
Example:
- Gas in a sealed container where heat can pass through the walls.
Most laboratory experiments involve closed systems.
Isolated System
An isolated system cannot exchange matter or energy with its surroundings.
Example:
- An ideal thermos flask (approximate isolated system)
In reality, perfectly isolated systems do not exist, but some systems closely approximate this condition.
2. Thermodynamic Properties
Thermodynamic properties describe the state of a system.
They are divided into two main categories.
Intensive Properties
Intensive properties do not depend on the amount of substance in the system.
Examples include:
- Temperature
- Pressure
- Density
These properties remain the same regardless of system size.
Extensive Properties
Extensive properties depend on the quantity of matter.
Examples include:
- Volume
- Mass
- Internal energy
If the system size doubles, these properties also double.
3. State Functions and Path Functions
Thermodynamics distinguishes between state functions and path functions.
State Functions
State functions depend only on the current state of the system, not on the path taken to reach that state.
Examples include:
- Internal energy
- Enthalpy
- Entropy
- Pressure
- Temperature
Path Functions
Path functions depend on the specific process used to change the system.
Examples include:
- Heat
- Work
These quantities vary depending on how the system moves from one state to another.
4. The Zeroth Law of Thermodynamics
The Zeroth Law establishes the concept of temperature.
It states:
If two systems are in thermal equilibrium with a third system, they are also in thermal equilibrium with each other.
This principle allows the use of thermometers to measure temperature.
Example:
If object A is in thermal equilibrium with object C, and object B is also in equilibrium with object C, then A and B must be at the same temperature.
This law forms the basis for temperature measurement.
5. The First Law of Thermodynamics
\Delta U = Q – W
Definition
The First Law of Thermodynamics is essentially the law of conservation of energy.
It states that energy cannot be created or destroyed; it can only be transferred or converted from one form to another.
Explanation
According to the first law, the change in internal energy of a system depends on:
- Heat added to the system
- Work done by the system
If heat is added, internal energy increases.
If work is done by the system, internal energy decreases.
Internal Energy
Internal energy is the total energy contained within a system.
It includes:
- Kinetic energy of molecules
- Potential energy from intermolecular interactions
Changes in internal energy occur during heating, cooling, compression, expansion, or chemical reactions.
Work in Thermodynamics
Work occurs when a force causes displacement.
In thermodynamics, work often occurs when gases expand or compress.
Example:
Gas expansion pushing a piston upward.
6. Heat and Energy Transfer
Heat is the transfer of energy between systems due to temperature differences.
There are three main mechanisms of heat transfer.
Conduction


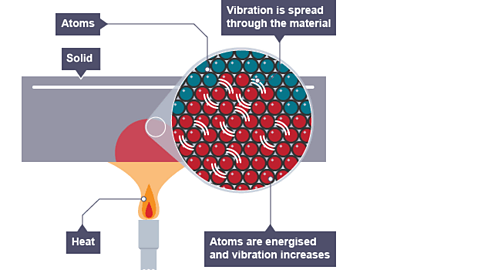
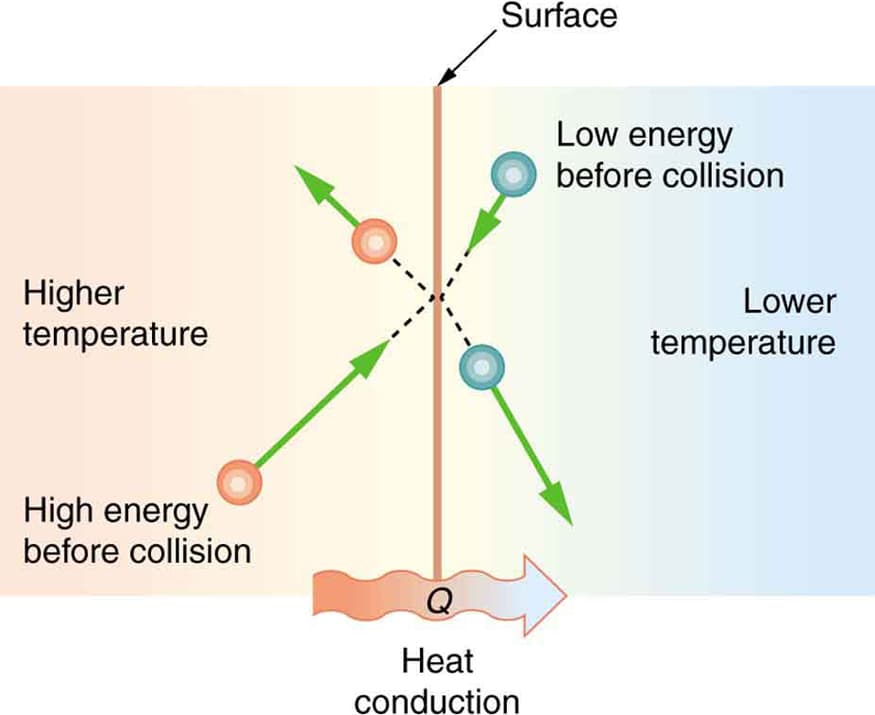
Conduction is the transfer of heat through direct contact between particles.
Example:
A metal spoon placed in hot tea becomes warm.
Heat flows from higher temperature to lower temperature regions.
Convection




Convection occurs in fluids (liquids and gases) when heat transfer occurs through fluid motion.
Examples include:
- Boiling water
- Atmospheric circulation
Warm fluid rises while cooler fluid sinks, creating convection currents.
Radiation




Radiation is heat transfer through electromagnetic waves.
Unlike conduction and convection, radiation does not require a medium.
Example:
Heat from the sun reaching Earth.
7. The Second Law of Thermodynamics
The Second Law introduces the concept of entropy.
It states that the entropy of an isolated system always increases over time.
In simpler terms, natural processes tend to move toward greater disorder.
Entropy
Entropy is a measure of randomness or disorder in a system.
Examples:
- Ice melting increases entropy.
- Gas expansion increases entropy.
Systems tend to move toward states with higher entropy.
Spontaneous Processes
A spontaneous process occurs naturally without external intervention.
Examples include:
- Heat flowing from hot to cold objects
- Mixing of gases
- Dissolution of salt in water
Spontaneous processes generally increase entropy.
8. The Third Law of Thermodynamics
The Third Law states that the entropy of a perfect crystal approaches zero as the temperature approaches absolute zero.
Absolute zero is the lowest possible temperature.
Absolute zero:
0 Kelvin
−273.15 °C
At this temperature, molecular motion theoretically stops.
9. Enthalpy
Enthalpy represents the heat content of a system.
It is particularly useful when studying reactions at constant pressure.
The change in enthalpy during a reaction is called enthalpy change.
Exothermic Reactions
Exothermic reactions release heat to the surroundings.
Examples:
- Combustion of fuels
- Respiration in living organisms
Endothermic Reactions
Endothermic reactions absorb heat from the surroundings.
Examples:
- Photosynthesis
- Melting of ice
10. Thermodynamic Processes
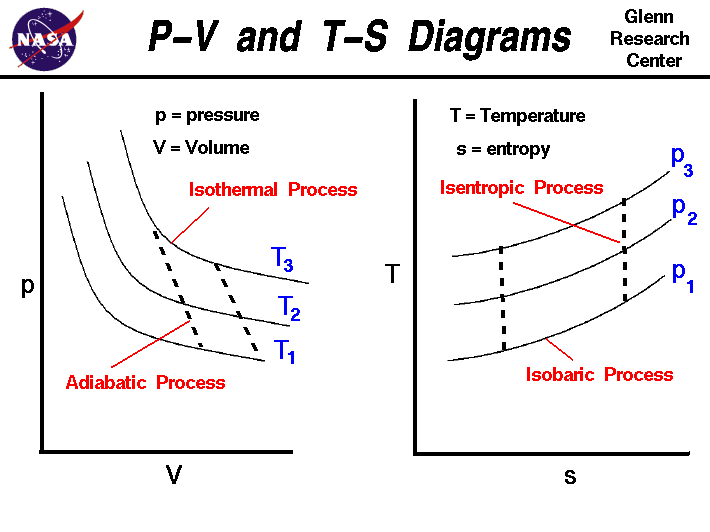



Thermodynamic processes describe how systems change from one state to another.
Isothermal Process
Temperature remains constant.
Heat transfer occurs to maintain constant temperature during expansion or compression.
Adiabatic Process
No heat exchange occurs between system and surroundings.
Energy changes occur only through work.
Isobaric Process
Pressure remains constant while volume and temperature may change.
Isochoric Process
Volume remains constant.
Heat transfer changes temperature and pressure.
11. Thermodynamic Cycles
Thermodynamic cycles occur when a system returns to its initial state after a series of processes.
Examples include:
- Carnot cycle
- Rankine cycle
- Otto cycle
These cycles are important in heat engines and power plants.
12. Applications of Thermodynamics
Thermodynamics is essential in many scientific and engineering fields.
Power Generation
Power plants convert thermal energy into mechanical energy using thermodynamic cycles.
Refrigeration
Refrigerators and air conditioners operate based on thermodynamic principles involving heat transfer and phase changes.
Chemical Engineering
Thermodynamics helps design reactors and optimize industrial chemical processes.
Biological Systems
Living organisms rely on thermodynamic principles for metabolism and energy transfer.
Environmental Science
Thermodynamics explains climate systems, atmospheric circulation, and energy balance on Earth.
13. Importance of Thermodynamics
Thermodynamics provides the fundamental framework for understanding energy transformations in nature and technology.
It explains how energy moves between systems, how chemical reactions release or absorb heat, and how engines convert heat into mechanical work.
The laws of thermodynamics apply universally, governing processes in physics, chemistry, biology, engineering, and environmental science.
Understanding thermodynamics enables scientists and engineers to design efficient machines, improve energy systems, and develop sustainable technologies.
Conclusion
Thermodynamics is the scientific study of energy, heat, and work and the laws that govern their transformations. It provides a powerful framework for understanding how energy flows through physical and chemical systems.
The four fundamental laws of thermodynamics describe the principles of temperature equilibrium, energy conservation, entropy increase, and the behavior of matter at extremely low temperatures. These laws apply universally to all physical processes.
Key thermodynamic concepts such as internal energy, enthalpy, entropy, and thermodynamic processes help explain the behavior of systems ranging from microscopic chemical reactions to large-scale industrial machines.
Thermodynamics plays a vital role in numerous applications, including power generation, refrigeration, chemical manufacturing, biological metabolism, and environmental systems. By studying thermodynamics, scientists gain insight into the fundamental principles governing energy transformations in the universe.