
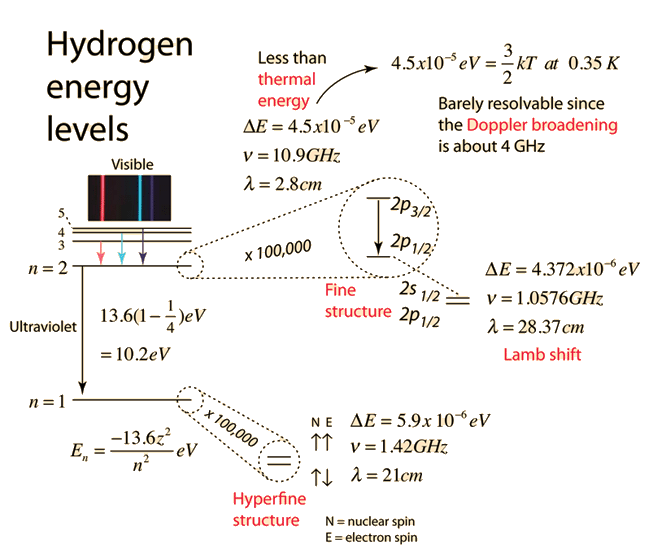
Introduction
Fine structure refers to the small splitting of spectral lines in the emission or absorption spectra of atoms. When atoms emit or absorb radiation, the spectral lines are not always single sharp lines. Instead, careful observation with high-resolution spectrometers shows that many spectral lines are split into closely spaced components. This phenomenon is known as fine structure.
Fine structure arises due to subtle interactions inside atoms, mainly involving electron spin, relativistic corrections to electron motion, and the interaction between electron spin and orbital angular momentum. These effects cause slight differences in energy levels that were previously considered identical in simpler atomic models.
The study of fine structure played an important role in the development of quantum mechanics and atomic theory. It helped scientists understand the internal dynamics of atoms and provided strong experimental evidence for the existence of electron spin and relativistic effects.
Important contributors to the understanding of fine structure include physicists such as Arnold Sommerfeld, Wolfgang Pauli, Paul Dirac, and Niels Bohr.
Fine structure is an essential concept in atomic spectroscopy, quantum mechanics, and astrophysics, providing insights into atomic energy levels and electron interactions.
Historical Background
Early Spectral Observations
In the late nineteenth and early twentieth centuries, scientists studying atomic spectra noticed that certain spectral lines were not single lines but consisted of several closely spaced components. These observations became possible with improvements in spectroscopic techniques.
Initially, atomic models such as the Bohr model predicted only single spectral lines corresponding to transitions between discrete energy levels. However, experimental measurements revealed small splittings in these lines that the Bohr model could not explain.
Sommerfeld’s Contribution
A major step toward explaining fine structure came from the work of Arnold Sommerfeld. Sommerfeld extended the Bohr model by introducing relativistic corrections and elliptical electron orbits.
His model allowed for additional quantum numbers and provided an explanation for the splitting of spectral lines observed in hydrogen and hydrogen-like atoms.
Although Sommerfeld’s model was later replaced by quantum mechanics, it successfully predicted several features of fine structure.
Causes of Fine Structure
Fine structure arises from three primary effects:
- Relativistic corrections to electron motion
- Spin–orbit interaction
- Quantum mechanical corrections
These effects cause slight changes in the energy levels of electrons within atoms.
Relativistic Corrections
Electrons in atoms move at very high speeds, particularly in atoms with large nuclear charge. When electron velocities approach a significant fraction of the speed of light, relativistic effects become important.
Relativistic mechanics modifies the relationship between momentum and energy. This leads to small corrections in the energy levels of electrons.
The relativistic correction term for energy is approximately proportional to:
[
E_{rel} \propto \frac{Z^4}{n^3}
]
where:
- (Z) = atomic number
- (n) = principal quantum number
This correction becomes more significant in heavier atoms.
Spin–Orbit Interaction


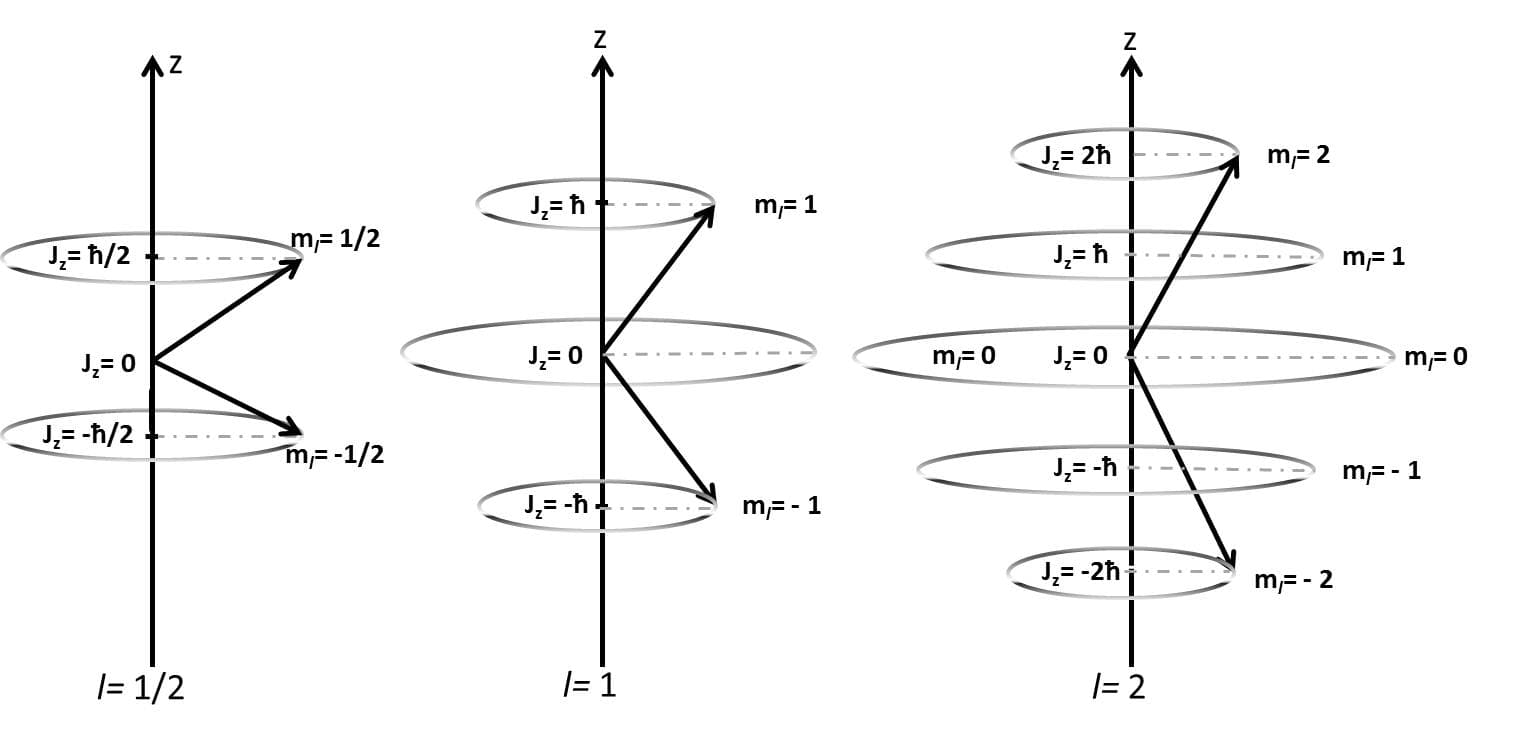

One of the most important causes of fine structure is spin–orbit interaction.
Spin–orbit coupling occurs because the electron has:
- Orbital angular momentum
- Intrinsic spin angular momentum
As the electron moves around the nucleus, it experiences a magnetic field created by its own orbital motion. The electron’s magnetic moment due to spin interacts with this magnetic field.
This interaction causes the energy levels to split into multiple sub-levels.
Total Angular Momentum
In the presence of spin–orbit interaction, the total angular momentum of the electron is given by:
[
\vec{J} = \vec{L} + \vec{S}
]
where:
- (L) = orbital angular momentum
- (S) = spin angular momentum
- (J) = total angular momentum
The quantum number (j) represents the magnitude of total angular momentum.
Possible values of (j) are:
[
j = l + s
]
[
j = l – s
]
Because (s = 1/2), each orbital splits into two energy levels.
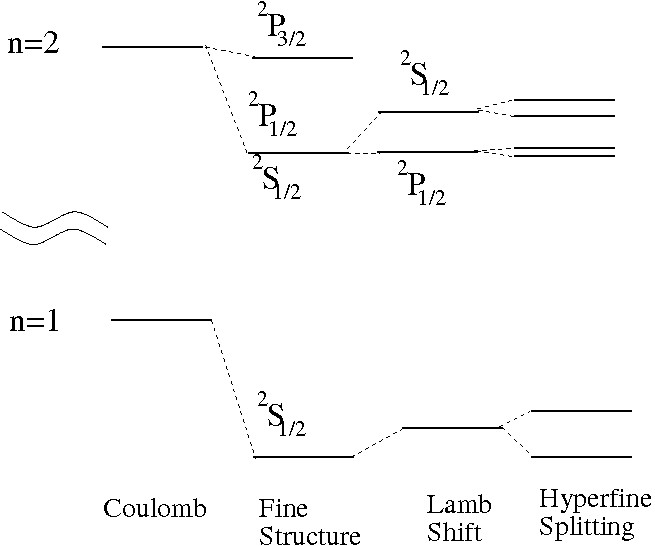
Fine Structure in Hydrogen

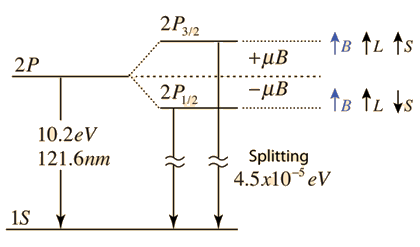


Hydrogen provides one of the simplest systems for studying fine structure.
In hydrogen, fine structure results from:
- Relativistic corrections
- Spin–orbit interaction
- Darwin term (quantum correction)
The corrected energy levels depend on both the principal quantum number (n) and the total angular momentum quantum number (j).
The energy expression becomes approximately:
[
E_{n,j} = -\frac{13.6}{n^2} \left[1 + \frac{\alpha^2}{n^2}\left(\frac{n}{j+1/2} – \frac{3}{4}\right)\right]
]
where:
- (\alpha) = fine-structure constant.
Fine-Structure Constant
The fine-structure constant is a fundamental dimensionless constant in physics.
It is represented by:
[
\alpha = \frac{e^2}{4\pi \varepsilon_0 \hbar c}
]
Its approximate value is:
[
\alpha \approx \frac{1}{137}
]
This constant determines the strength of electromagnetic interactions and plays a key role in fine structure.
Dirac Theory and Fine Structure
A more complete explanation of fine structure was provided by the relativistic quantum theory developed by Paul Dirac.
The Dirac equation combines quantum mechanics with special relativity and naturally includes electron spin.
One of the major achievements of the Dirac equation was its ability to predict fine structure in hydrogen without additional assumptions.
Spectral Line Splitting
Fine structure causes spectral lines to split into several closely spaced lines.
These lines correspond to transitions between slightly different energy levels.
For example:
A single spectral line predicted by the Bohr model may appear as two or more lines when observed with high-resolution spectroscopy.
Selection Rules for Fine Structure
Quantum mechanical selection rules determine which transitions between energy levels are allowed.
Common rules include:
[
\Delta l = \pm 1
]
[
\Delta j = 0, \pm 1
]
Transitions violating these rules are forbidden or highly unlikely.
Comparison with Hyperfine Structure
Fine structure should not be confused with hyperfine structure.
Fine structure results from interactions involving the electron itself.
Hyperfine structure arises from interactions between the electron and the nucleus, particularly nuclear spin.
Hyperfine splitting is typically much smaller than fine structure splitting.
Experimental Observation
Fine structure is observed using high-resolution spectroscopic techniques.
In laboratories, scientists use:
- Diffraction gratings
- Interferometers
- Laser spectroscopy
These instruments allow precise measurement of small wavelength differences.
Zeeman Effect and Fine Structure
Fine structure can be further modified in the presence of a magnetic field.
This leads to the Zeeman effect, discovered by Pieter Zeeman.
The Zeeman effect causes additional splitting of spectral lines due to interaction between magnetic fields and atomic magnetic moments.
Stark Effect
Another phenomenon related to spectral splitting is the Stark effect, discovered by Johannes Stark.
The Stark effect occurs when spectral lines split under the influence of an external electric field.
Both the Zeeman and Stark effects provide valuable information about atomic structure.
Fine Structure in Multi-Electron Atoms
In atoms with more than one electron, fine structure becomes more complex.
Additional interactions occur between electrons, including:
- Electron–electron repulsion
- Exchange interactions
- Spin coupling
These interactions lead to more complicated energy level structures.
Importance in Atomic Physics
Fine structure provides crucial insights into atomic structure and electron behavior.
It helps scientists understand:
- Electron angular momentum
- Spin interactions
- Relativistic effects
- Quantum mechanical energy corrections
Fine structure measurements are used to test theoretical predictions in atomic physics.
Applications of Fine Structure
Fine structure plays an important role in many scientific fields.
Spectroscopy
Fine structure analysis helps identify elements and study atomic transitions.
Astrophysics
Astronomers analyze fine structure lines to determine the physical conditions in stars and interstellar clouds.
Fundamental Physics
Precise measurements of fine structure allow scientists to test quantum electrodynamics and measure fundamental constants.
Laser Physics
Fine structure transitions are used in certain types of lasers.
Modern Research
Modern research in atomic physics continues to study fine structure using advanced experimental techniques.
High-precision spectroscopy allows scientists to measure extremely small energy differences and test theoretical predictions with great accuracy.
Fine structure studies also contribute to research in:
- Quantum electrodynamics
- Atomic clocks
- Precision measurements of fundamental constants.
Conclusion
Fine structure represents a subtle but important feature of atomic spectra. It arises from relativistic corrections, spin–orbit interaction, and quantum mechanical effects within atoms. These interactions cause slight splitting of energy levels, which leads to the splitting of spectral lines observed in atomic spectra.
The study of fine structure played a significant role in the development of quantum mechanics and provided strong evidence for the existence of electron spin and relativistic effects in atomic systems. Advances in spectroscopy have allowed scientists to measure fine structure with remarkable precision, providing valuable insights into atomic structure and fundamental physical laws.
Today, fine structure continues to be an important topic in atomic physics, spectroscopy, and astrophysics, helping scientists explore the behavior of atoms and the fundamental interactions that govern the microscopic world.
