
Introduction
The Second Law of Thermodynamics is one of the most important principles in physics and engineering. It describes the direction of natural processes and the concept of entropy, explaining why certain processes occur spontaneously while others do not.
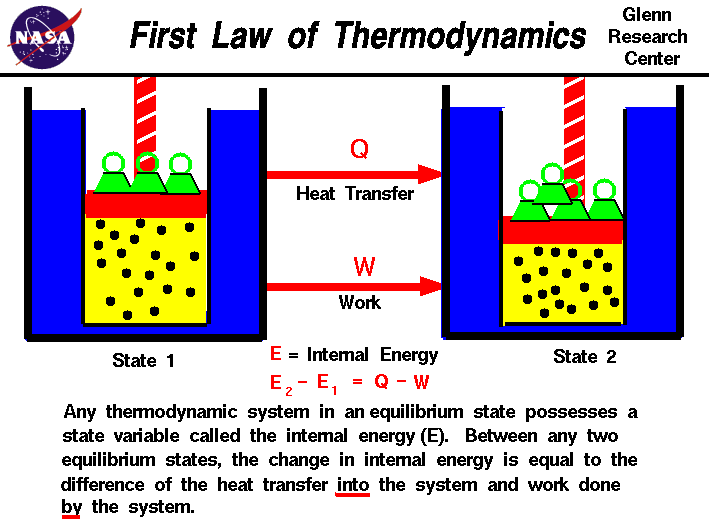
While the First Law of Thermodynamics states that energy is conserved, the second law explains how energy flows and why energy transformations are not perfectly efficient.
In simple terms, the second law states that:
Heat naturally flows from a hotter body to a colder body, and the total entropy of an isolated system always increases over time.
This law introduces the concept of irreversibility in natural processes and explains why energy conversions always involve some loss of useful energy.
The second law is fundamental to understanding:
- Heat engines
- Refrigerators and air conditioners
- Chemical reactions
- Biological processes
- Energy efficiency
- The direction of time in thermodynamics
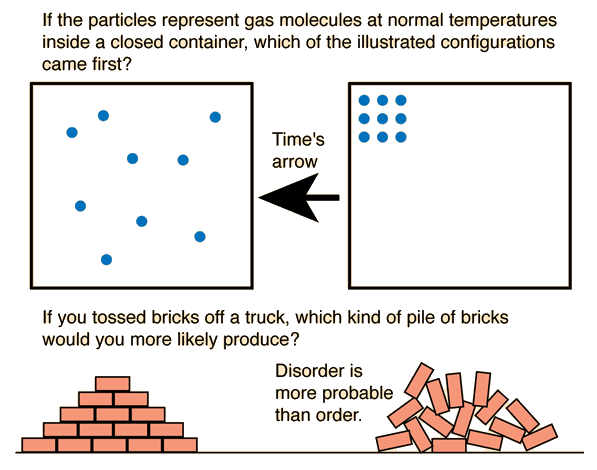
It also introduces the concept of entropy, which measures the level of disorder or randomness in a system.
Historical Development




The second law emerged during the Industrial Revolution, when scientists tried to understand how efficiently heat engines could convert heat into mechanical work.
Important contributors include:
Sadi Carnot (1824)
Carnot studied heat engines and proposed the Carnot cycle, which represents the most efficient possible heat engine.
Rudolf Clausius
Clausius introduced the concept of entropy and expressed the second law mathematically.
Lord Kelvin (William Thomson)
Kelvin provided another formulation of the second law regarding the impossibility of converting heat completely into work.
These scientists showed that energy transformations have natural limits, and perfect efficiency is impossible.
Fundamental Statements of the Second Law




There are two major statements of the second law.
Kelvin–Planck Statement
The Kelvin–Planck statement focuses on heat engines.
It states:
It is impossible to construct a heat engine that converts all the heat absorbed from a reservoir into useful work.
This means that no heat engine can have 100% efficiency.
Some heat must always be rejected to a colder reservoir.
Example
In a car engine:
- Heat from fuel combustion produces work.
- Some heat is lost through exhaust gases and cooling systems.
Clausius Statement
The Clausius statement focuses on heat transfer.
It states:
Heat cannot spontaneously flow from a colder body to a hotter body without external work.
Example
A refrigerator transfers heat from inside the fridge (cold region) to the kitchen (warm region), but it requires electric power to do so.
Entropy – The Key Concept




The most important concept introduced by the second law is entropy.
Definition
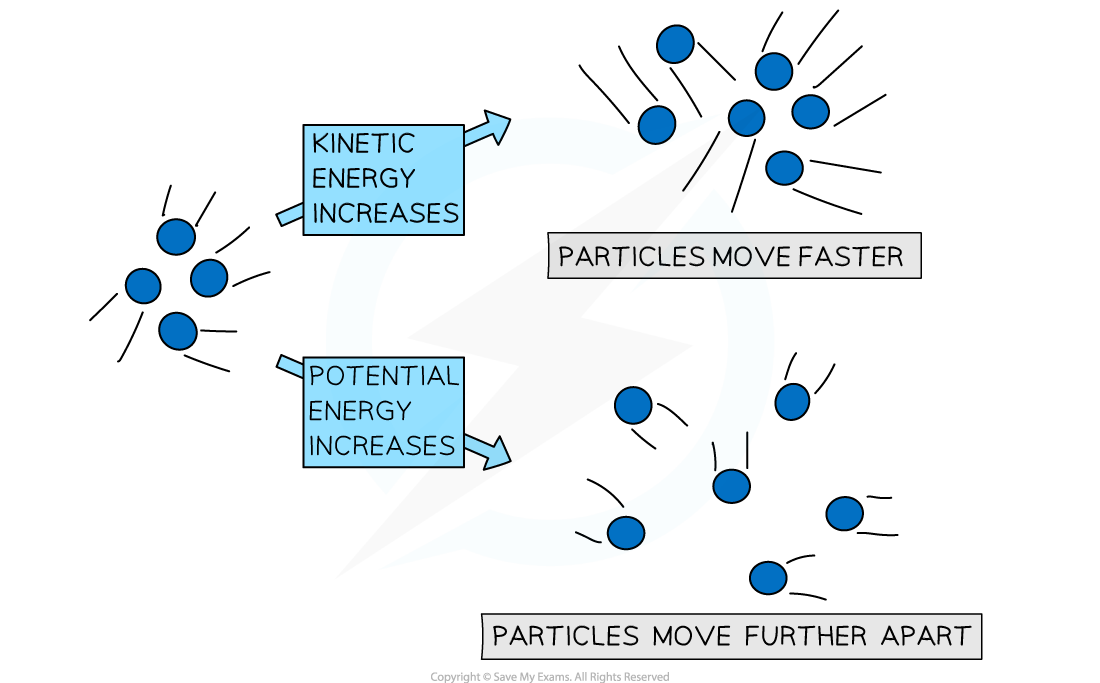
Entropy is a measure of the disorder or randomness of a system.
Symbol:
[
S
]
Units:
[
J/K
]
Entropy Change Equation
[
\Delta S = \frac{Q}{T}
]
Where:
- ( \Delta S ) = Change in entropy
- ( Q ) = Heat transfer
- ( T ) = Absolute temperature
Understanding Entropy
Systems naturally move from order to disorder.
Examples:
- Ice melting into water
- Perfume spreading in air
- Gas expanding into empty space
These processes increase entropy.
Entropy and Microstates
In statistical mechanics, entropy is related to the number of possible microscopic arrangements.
[
S = k \ln W
]
Where:
- ( S ) = Entropy
- ( k ) = Boltzmann constant
- ( W ) = Number of microstates
More microstates → higher entropy.
Reversible and Irreversible Processes
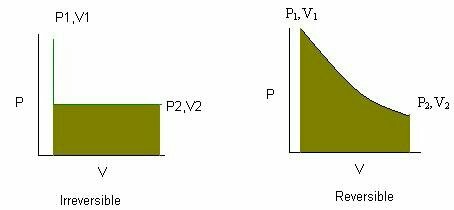
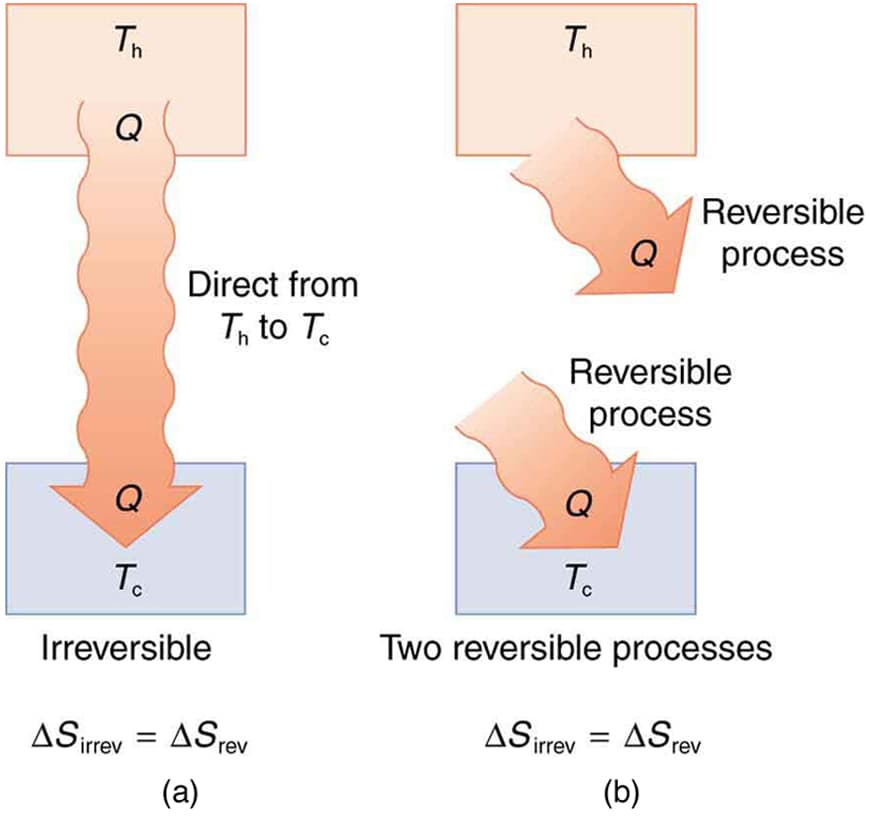
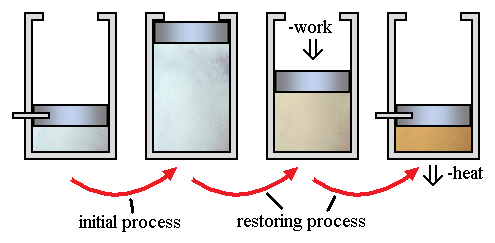

Thermodynamic processes are classified as reversible or irreversible.
Reversible Process
A reversible process is an ideal process that occurs infinitely slowly and can be reversed without energy loss.
Characteristics:
- No friction
- No turbulence
- System remains in equilibrium
Example:
Ideal gas expansion in a perfectly controlled environment.
Irreversible Process
Most natural processes are irreversible.
Characteristics:
- Energy dissipation
- Friction
- Turbulence
- Heat loss
Examples:
- Combustion
- Mixing of gases
- Heat transfer across temperature difference
Irreversible processes increase entropy.
Entropy Change in Different Processes
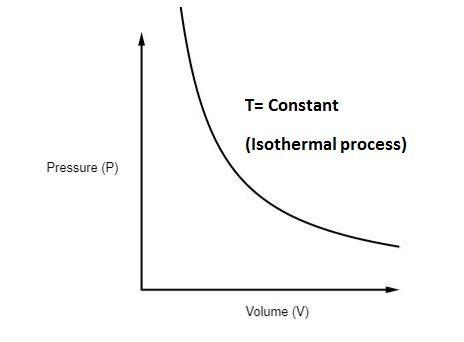
Isothermal Process
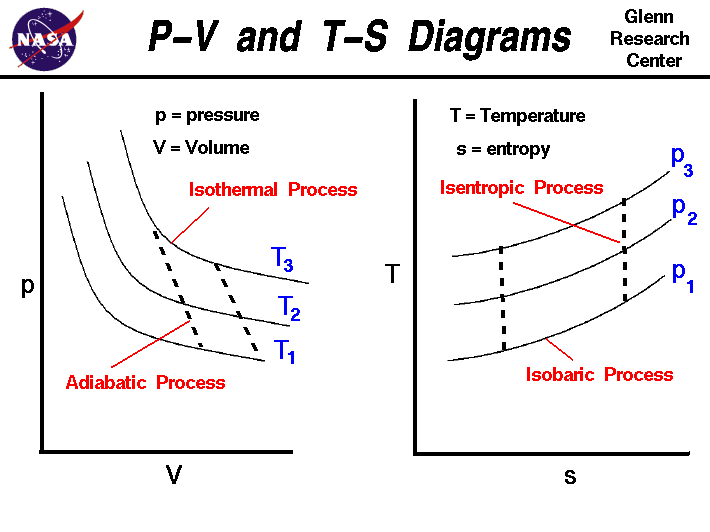


In an isothermal process, temperature remains constant.
Entropy change:
[
\Delta S = nR \ln \frac{V_2}{V_1}
]
Where:
- (n) = number of moles
- (R) = gas constant
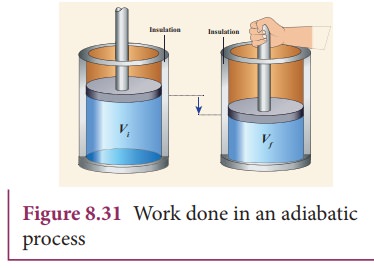
Adiabatic Process




In an ideal adiabatic process:
[
Q = 0
]
If the process is reversible:
[
\Delta S = 0
]
Such a process is called isentropic.
Phase Change



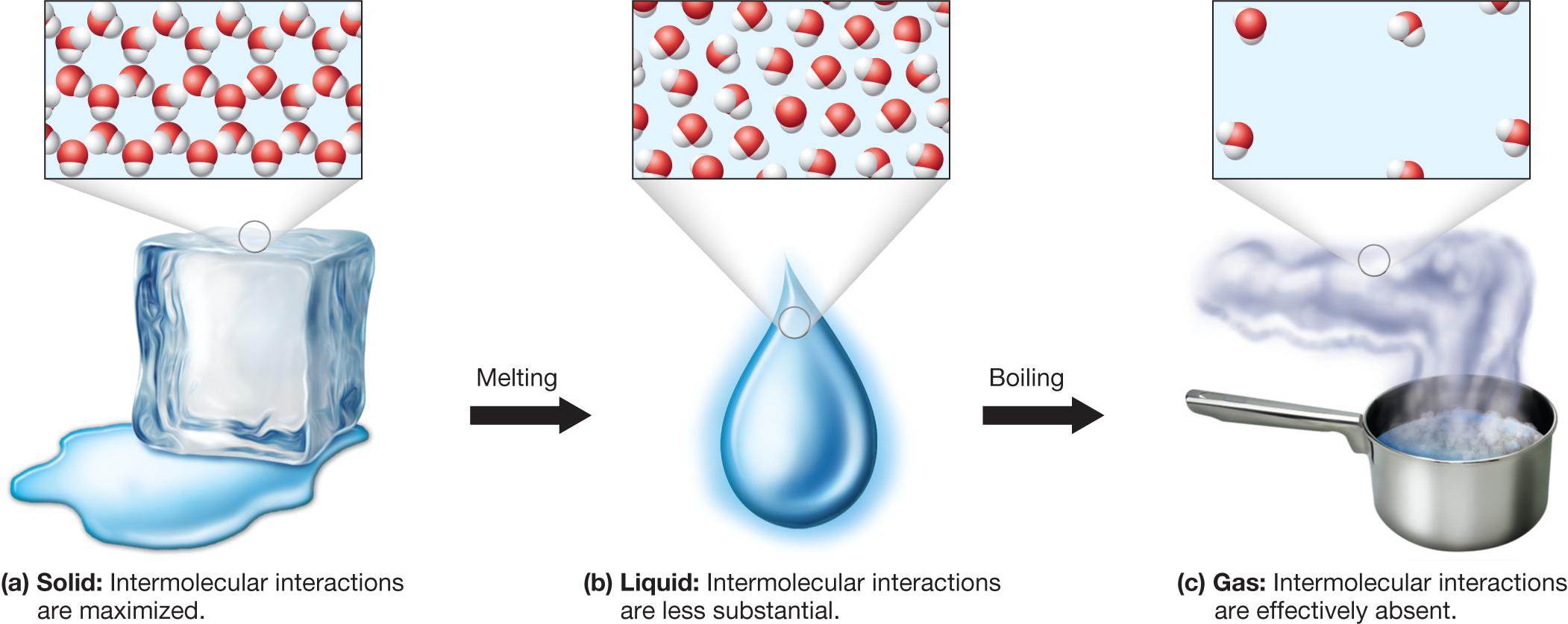
During phase changes, entropy usually increases.
Examples:
Solid → Liquid → Gas
Entropy increases because molecular freedom increases.
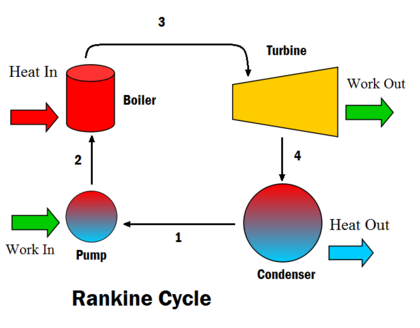
Heat Engines and the Second Law




A heat engine converts heat energy into mechanical work.
Components:
- Hot reservoir
- Working substance
- Cold reservoir
Efficiency
Efficiency is defined as:
[
\eta = \frac{W}{Q_H}
]
Where:
- (W) = Work output
- (Q_H) = Heat absorbed
Because of the second law:
[
\eta < 100%
]
Carnot Cycle


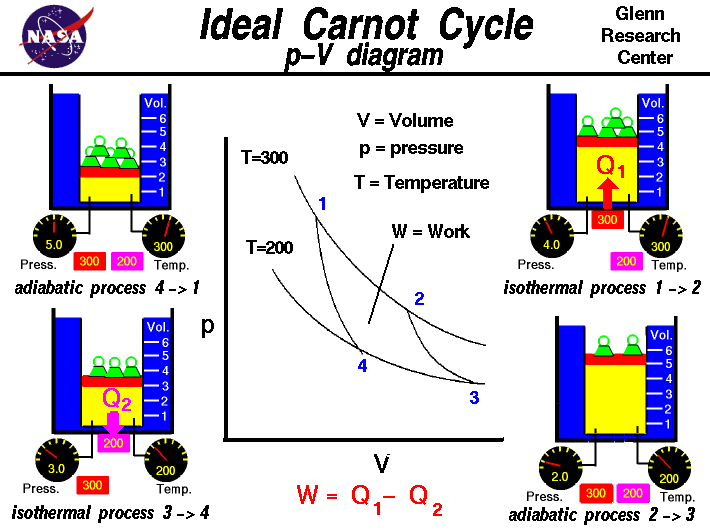
The Carnot cycle represents the most efficient heat engine possible.
It consists of four stages:
- Isothermal expansion
- Adiabatic expansion
- Isothermal compression
- Adiabatic compression
Carnot Efficiency
[
\eta = 1 – \frac{T_C}{T_H}
]
Where:
- (T_H) = Temperature of hot reservoir
- (T_C) = Temperature of cold reservoir
No real engine can exceed Carnot efficiency.
Refrigerators and Heat Pumps




A refrigerator transfers heat from a cold space to a warmer space using external work.
Coefficient of Performance (COP)
[
COP = \frac{Q_C}{W}
]
Where:
- (Q_C) = Heat removed from cold region
- (W) = Work input
Heat pumps operate similarly but are used for heating buildings.
Entropy and the Arrow of Time

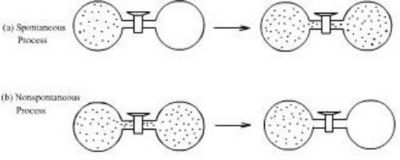
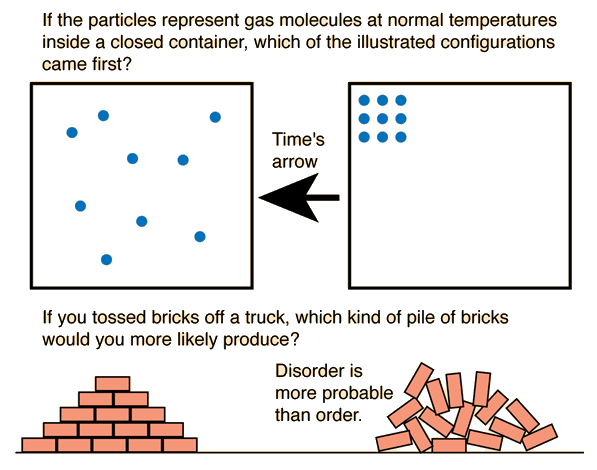

The second law explains the arrow of time.
Natural processes move in the direction of increasing entropy.
Examples:
- A broken cup cannot reassemble spontaneously.
- Heat flows from hot to cold.
- Gases mix rather than separate.
This explains why time appears to move in one direction.
Applications of the Second Law




The second law is used in many fields.
Engineering
- Engine design
- Turbines
- Power plants
Chemistry
- Chemical reaction spontaneity
- Thermodynamic equilibrium
Biology
Living organisms maintain order by exporting entropy to surroundings.
Environmental Science
Energy efficiency and sustainability analysis rely on second-law principles.
Limitations of Energy Conversion
The second law explains why:
- Perpetual motion machines are impossible
- Perfect energy efficiency cannot exist
- Waste heat is unavoidable in machines
Energy quality decreases during transformations.
High-quality energy (like electricity) often becomes low-quality energy (heat).
Importance of the Second Law
The Second Law is essential because it:
- Explains natural process direction
- Introduces entropy
- Sets limits on machine efficiency
- Explains irreversibility in nature
- Provides foundation for statistical mechanics
- Helps understand energy degradation
It also plays a key role in cosmology and understanding the evolution of the universe.
Conclusion
The Second Law of Thermodynamics describes the natural direction of energy transformations and introduces the concept of entropy, which measures disorder in a system.
While the first law states that energy is conserved, the second law explains why energy conversions are never perfectly efficient and why some processes occur spontaneously while others do not.
The law has profound implications in physics, chemistry, engineering, biology, and environmental science. It governs the operation of heat engines, refrigeration systems, and power plants, and explains the fundamental nature of time and irreversibility in the universe.
By understanding the second law, scientists and engineers can design more efficient technologies and better understand the behavior of natural systems.