


Introduction
The First Law of Thermodynamics is one of the fundamental principles of physics and thermodynamics that describes the conservation of energy in thermodynamic systems. It states that energy cannot be created or destroyed; instead, it can only be transferred or transformed from one form to another.
This law provides the foundation for understanding how energy moves between systems and their surroundings in the form of heat and work. It explains many natural phenomena such as heating, cooling, mechanical work, engine operation, and chemical reactions.
Thermodynamics studies the relationship between heat, work, temperature, and energy. The first law specifically deals with the energy balance of a system and shows how energy changes during physical and chemical processes.
Historically, the concept evolved during the 19th century when scientists such as James Prescott Joule, Julius Robert Mayer, and Hermann von Helmholtz demonstrated experimentally that mechanical work and heat are interchangeable forms of energy.
The First Law of Thermodynamics is widely used in many fields, including:
- Physics
- Chemistry
- Mechanical engineering
- Chemical engineering
- Environmental science
- Aerospace engineering
- Energy systems
Understanding this law allows scientists and engineers to design machines, engines, refrigerators, and power plants.
Basic Concepts of Thermodynamics



Before understanding the first law, several important thermodynamic concepts must be understood.
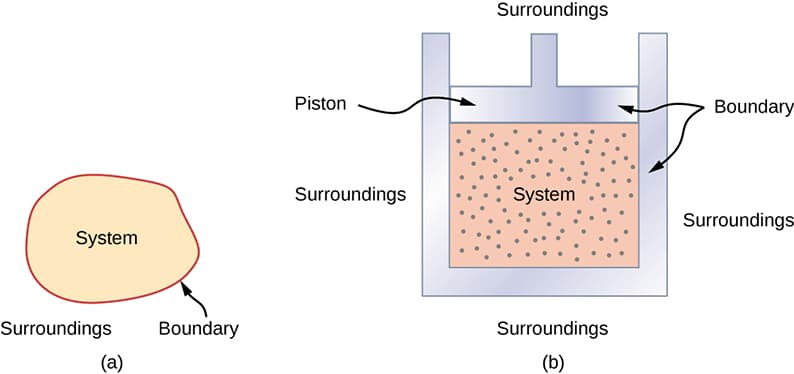
System
A thermodynamic system is a specific portion of the universe chosen for study.
Examples include:
- Gas inside a cylinder
- Water inside a boiler
- A chemical reaction mixture
- The Earth’s atmosphere
Everything outside the system is called the surroundings.
Types of Systems
Open System
An open system exchanges both energy and matter with the surroundings.
Examples:
- Steam turbine
- Human body
- Flowing river
Closed System
A closed system exchanges energy but not matter with surroundings.
Example:
- Gas inside a sealed piston-cylinder device.
Isolated System
An isolated system exchanges neither energy nor matter.
Example:
- The universe (considered an isolated system).
Boundary
The boundary separates the system from the surroundings. It may be real or imaginary.
Energy in Thermodynamics




Energy is the capacity to do work.
In thermodynamics, energy appears in different forms:
Kinetic Energy
Energy due to motion.
Example:
Moving gas molecules.
Potential Energy
Energy stored due to position or configuration.
Example:
Water stored in a dam.
Thermal Energy
Energy associated with temperature.
Chemical Energy
Energy stored in chemical bonds.
Electrical Energy
Energy due to electric charges.
Nuclear Energy
Energy stored in atomic nuclei.
All these forms of energy can transform into each other.
Internal Energy



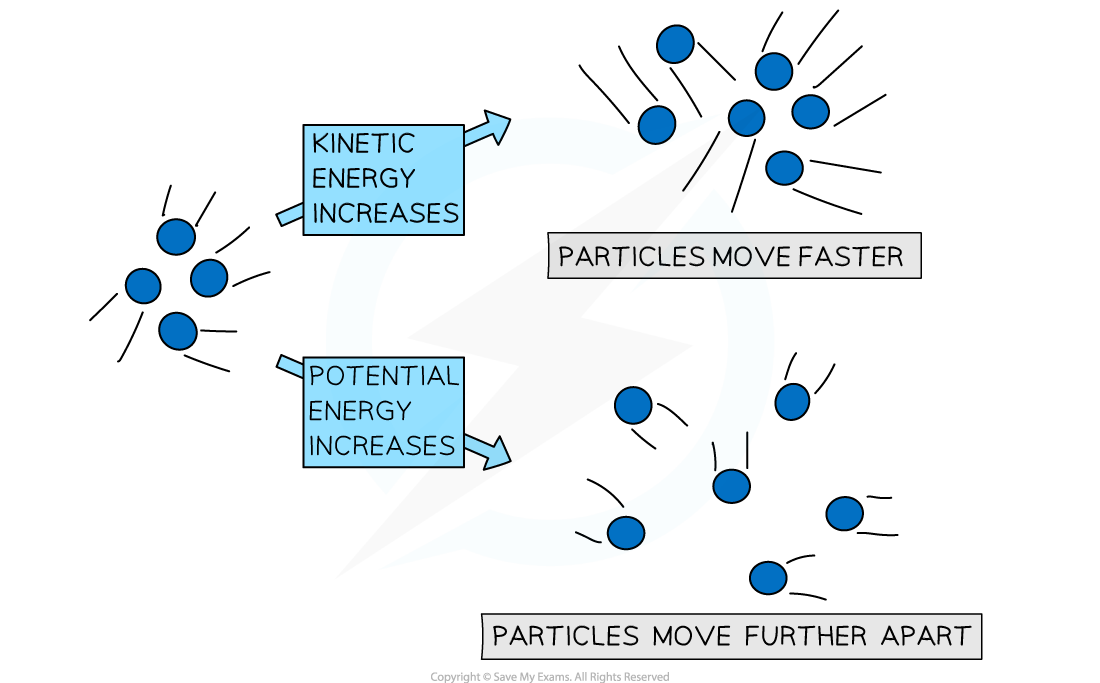
Internal energy (U) is the total microscopic energy contained within a system.
It includes:
- Kinetic energy of molecules
- Potential energy between molecules
- Rotational and vibrational energies
Internal energy depends mainly on temperature and state of the system.
Important points:
- Internal energy is a state function.
- It depends only on the current state of the system, not on the path taken to reach that state.
Heat and Work



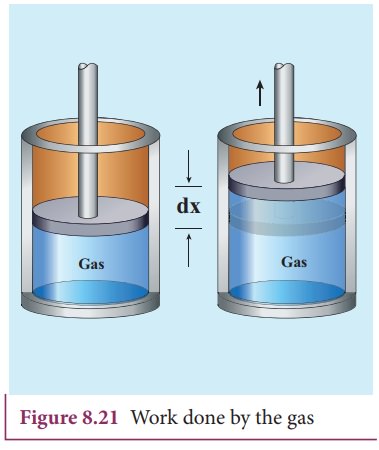
Energy can cross the system boundary in two ways:
Heat (Q)
Heat is energy transferred due to temperature difference between system and surroundings.
Examples:
- Heating water on a stove
- Sun warming the Earth
- Heat transfer in engines
Characteristics of Heat
- Flows from high temperature to low temperature
- Measured in joules (J) or calories
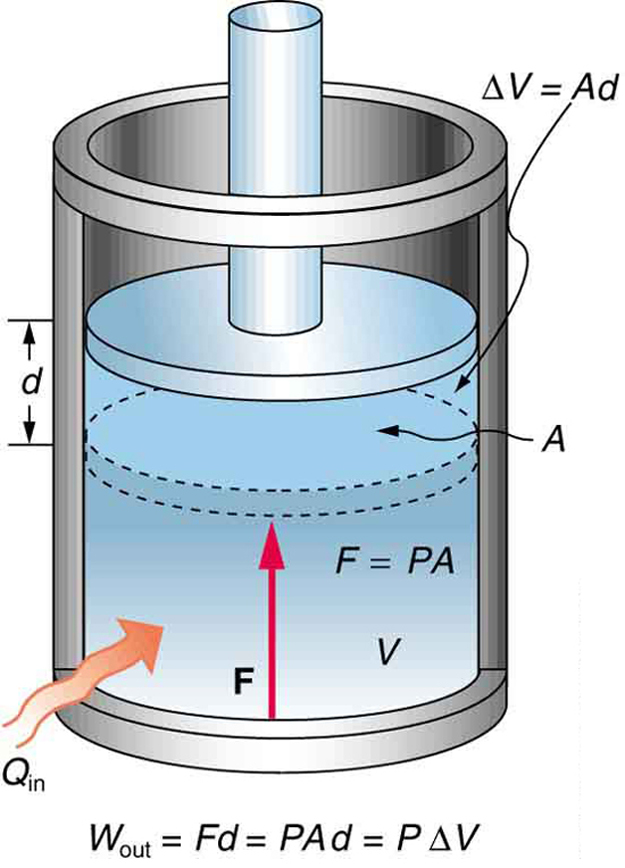
Work (W)
Work is energy transferred when a force causes displacement.
In thermodynamics, work commonly occurs when gas expands or compresses inside a piston.
Examples:
- Steam pushing a piston
- Air expanding in engines
Statement of the First Law of Thermodynamics


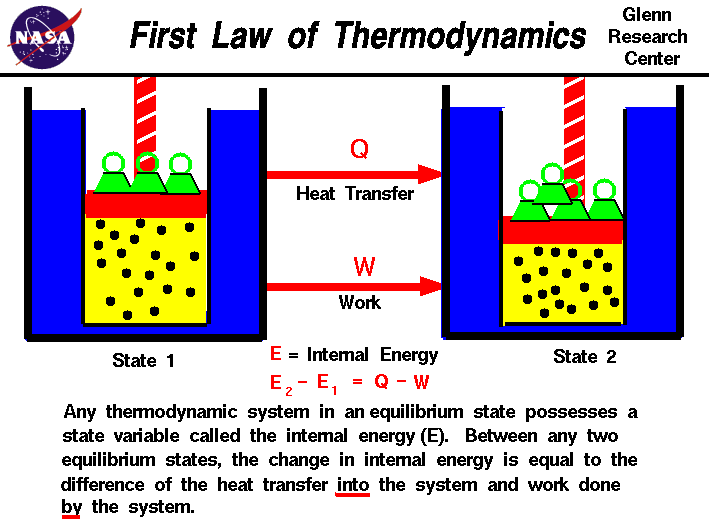

The First Law of Thermodynamics states:
The change in internal energy of a system equals the heat added to the system minus the work done by the system.
Mathematical Form
[
\Delta U = Q – W
]
Where:
- ( \Delta U ) = Change in internal energy
- ( Q ) = Heat supplied to the system
- ( W ) = Work done by the system
Interpretation
- If heat enters the system → internal energy increases
- If the system performs work → internal energy decreases
Sign Conventions
Understanding sign conventions is important in thermodynamics.
| Quantity | Sign | Meaning |
|---|---|---|
| Heat added to system | Positive | Energy enters system |
| Heat removed | Negative | Energy leaves system |
| Work done by system | Positive | System uses energy |
| Work done on system | Negative | Energy added to system |
First Law for Different Thermodynamic Processes
Isothermal Process
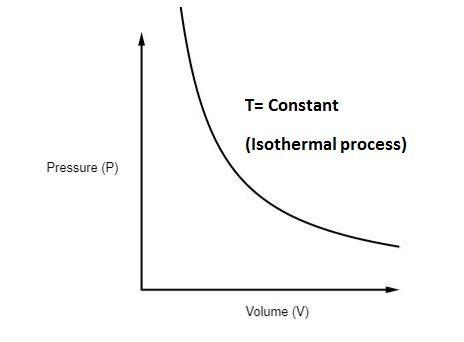



An isothermal process occurs at constant temperature.
Since internal energy depends on temperature:
[
\Delta U = 0
]
Thus,
[
Q = W
]
Meaning heat added to the system is completely converted into work.
Example:
Slow expansion of gas in a piston while temperature remains constant.
Adiabatic Process
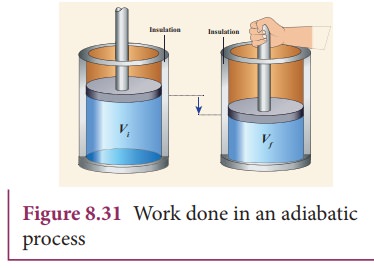
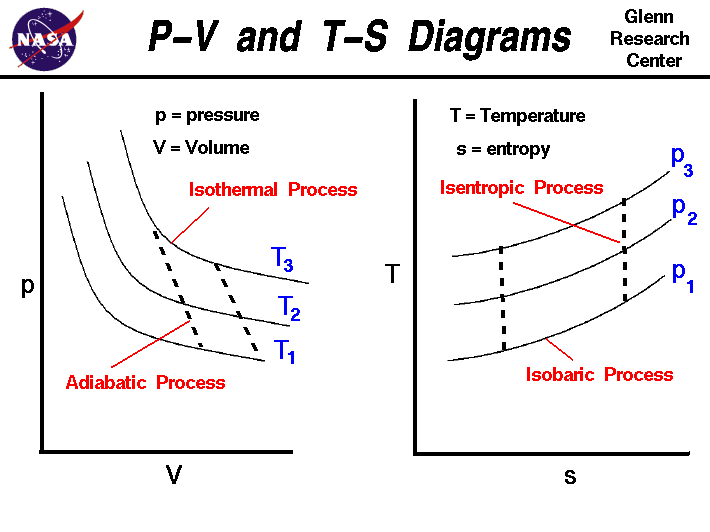


In an adiabatic process, no heat is exchanged with surroundings.
[
Q = 0
]
Thus:
[
\Delta U = -W
]
Internal energy changes due to work done.
Examples:
- Rapid compression of gas
- Atmospheric processes
- Diesel engine compression
Isochoric Process

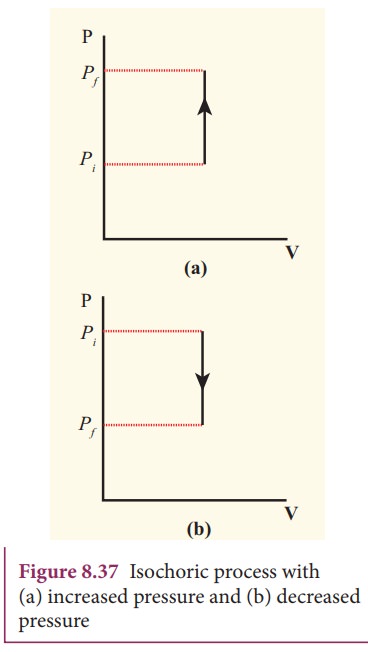

In an isochoric process, volume remains constant.
[
W = 0
]
Therefore:
[
\Delta U = Q
]
Heat added directly changes internal energy.
Example:
Heating gas in a rigid container.
Isobaric Process




In an isobaric process, pressure remains constant.
Work done:
[
W = P(V_2 – V_1)
]
The heat added is partly used for:
- Increasing internal energy
- Doing work
Applications of the First Law of Thermodynamics



The First Law has many practical applications.
Heat Engines
Heat engines convert thermal energy into mechanical work.
Examples:
- Steam engines
- Car engines
- Gas turbines
Refrigerators
Refrigerators use work to transfer heat from cold regions to warm surroundings.
Power Plants
Thermal power plants convert heat from fuel into electricity.
Biological Systems
Human metabolism follows energy conservation principles.
Limitations of the First Law
The First Law has some limitations.
It does not explain:
- Direction of heat flow
- Why heat flows from hot to cold
- Efficiency limits of engines
These questions are addressed by the Second Law of Thermodynamics.
Importance of the First Law
The First Law is one of the most important scientific principles because it:
- Establishes energy conservation
- Connects mechanical and thermal energy
- Forms the basis of thermodynamic analysis
- Helps design engines, turbines, and refrigerators
- Explains chemical energy transformations
Conclusion
The First Law of Thermodynamics expresses the fundamental principle of energy conservation in thermodynamic systems. It states that energy can neither be created nor destroyed but can only be transformed between different forms such as heat, work, and internal energy.
The law provides a mathematical relationship that allows scientists and engineers to analyze energy changes in physical and chemical processes. By understanding how heat and work influence internal energy, we can design machines, engines, power plants, and refrigeration systems that operate efficiently.
Although the First Law does not explain the direction of energy flow or the efficiency limits of energy conversion, it remains the foundation of thermodynamics and energy science.
