



Introduction
Radioactivity is the spontaneous emission of radiation from unstable atomic nuclei. It occurs when the nucleus of an atom undergoes a transformation in order to achieve a more stable configuration. During this process, the nucleus releases energy in the form of particles or electromagnetic radiation.
Radioactivity is a fundamental phenomenon in nuclear physics and plays a crucial role in many natural and technological processes. It occurs naturally in certain elements found in the Earth’s crust, as well as artificially in laboratories and nuclear reactors.
The discovery of radioactivity marked a major milestone in the development of modern physics. The phenomenon was first discovered in 1896 by Henri Becquerel while studying uranium salts. Later, extensive research by Marie Curie and Pierre Curie led to the discovery of additional radioactive elements such as radium and polonium.
Radioactivity revealed that atoms are not always stable and can undergo transformations that change their internal structure. The study of radioactive processes has led to important developments in nuclear energy, medicine, geology, and astrophysics.
Nature of Radioactivity
Radioactivity arises from the instability of atomic nuclei. A nucleus is composed of protons and neutrons, collectively known as nucleons. The stability of the nucleus depends on the balance between the attractive strong nuclear force and the repulsive electrostatic force between protons.
When this balance is disturbed, the nucleus becomes unstable and may undergo radioactive decay to reach a more stable state.
The radioactive decay process involves the emission of one or more of the following:
- Alpha particles
- Beta particles
- Gamma rays
These emissions carry away excess energy from the nucleus.
Discovery of Radioactivity
The discovery of radioactivity occurred accidentally during experiments with phosphorescent materials.
Henri Becquerel observed that uranium salts emitted radiation that could expose photographic plates even in the absence of sunlight.
This unexpected observation revealed that certain elements emit radiation spontaneously.
Later, Marie Curie and Pierre Curie conducted extensive research on radioactive materials and discovered new radioactive elements.
Their work laid the foundation for the field of nuclear physics.
Types of Radioactive Emissions
Radioactive decay produces three main types of radiation.
Alpha Radiation

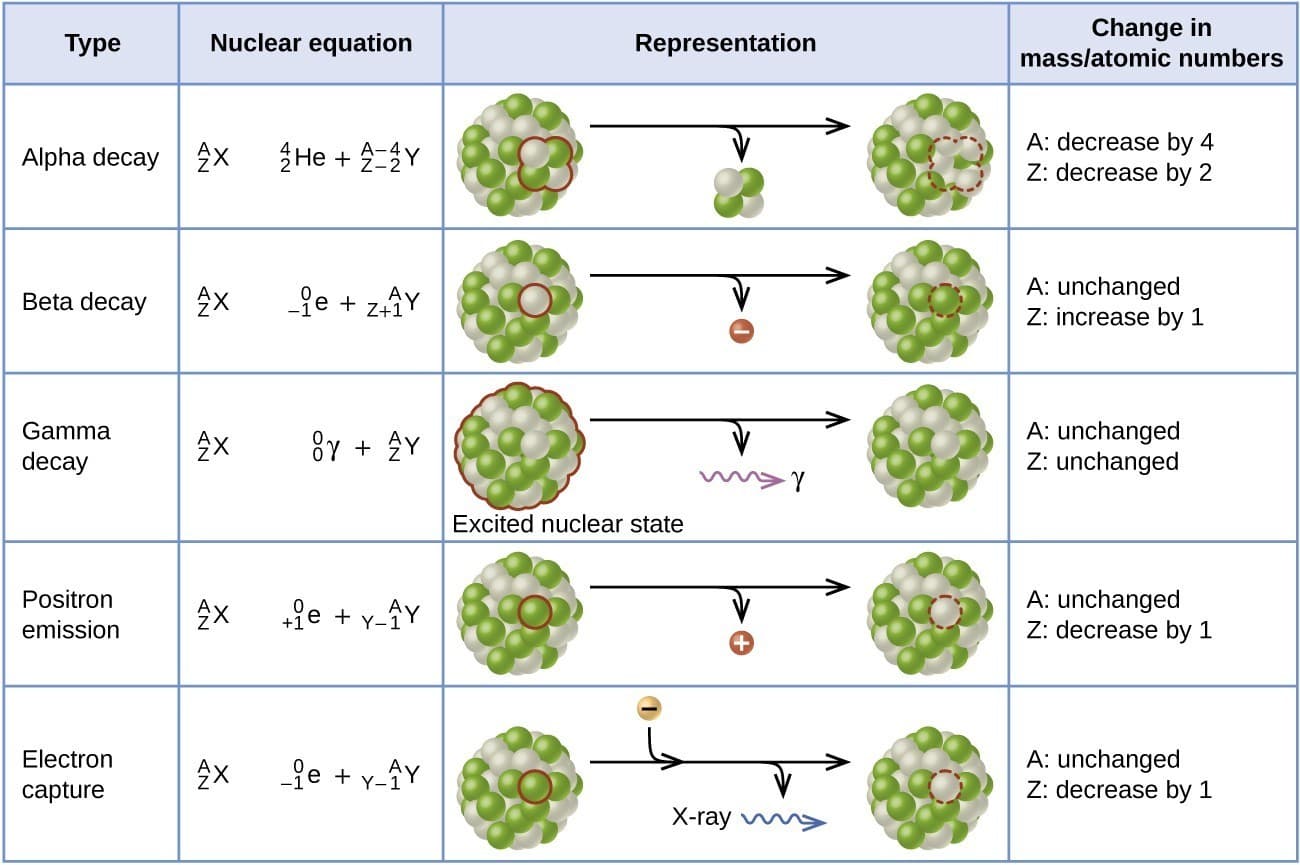
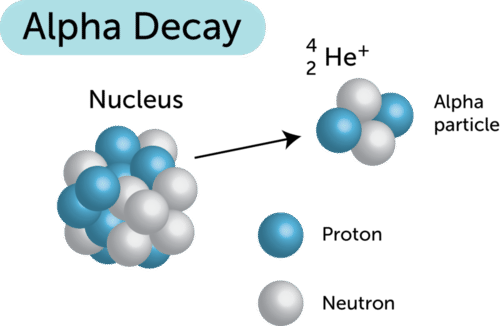
Alpha radiation consists of alpha particles, which are helium nuclei.
An alpha particle contains:
- 2 protons
- 2 neutrons
The nuclear reaction for alpha decay can be written as
[
^A_ZX \rightarrow ^{A-4}_{Z-2}Y + ^4_2He
]
Characteristics of alpha radiation:
- High mass
- Positive charge
- Low penetration power
- Strong ionizing ability
Alpha particles can be stopped by a sheet of paper or human skin.
Beta Radiation
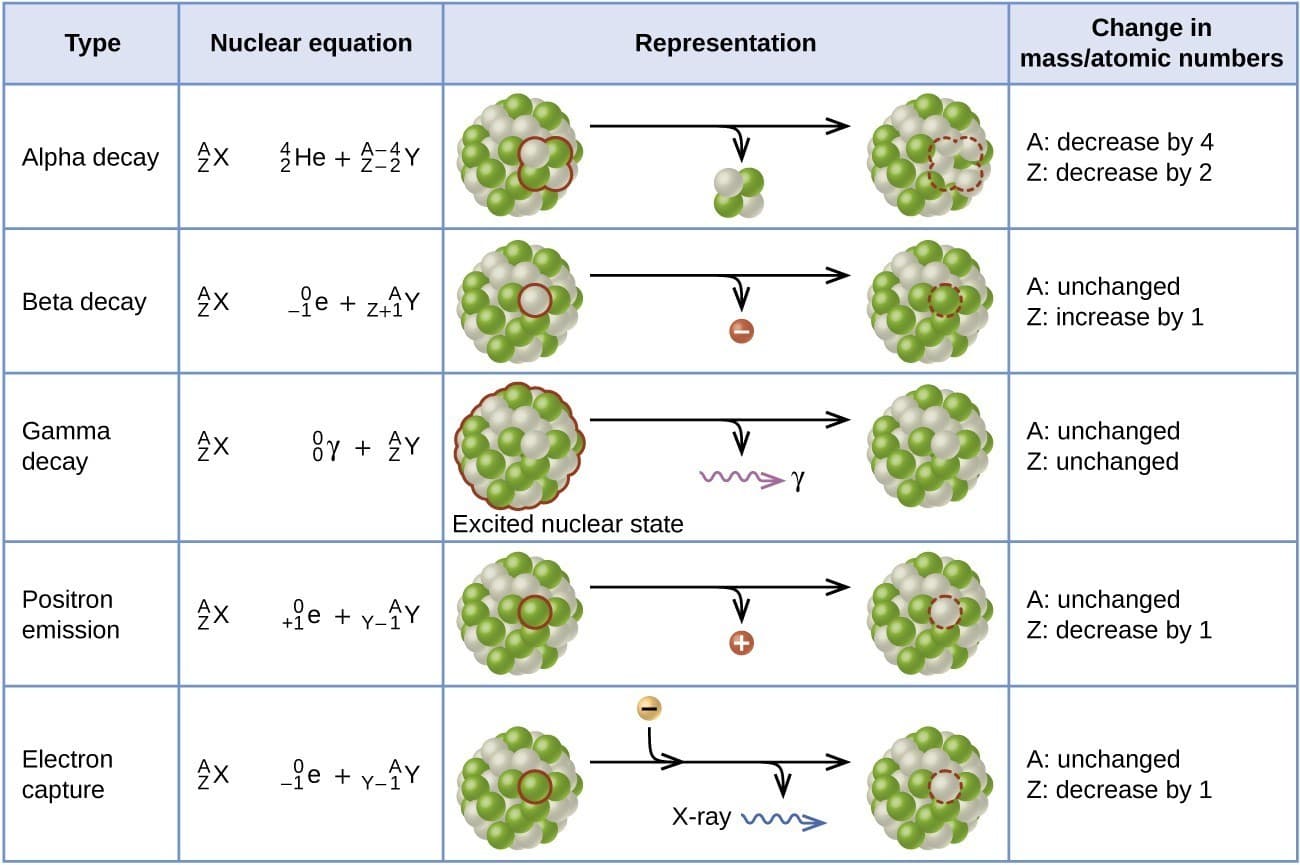



Beta radiation consists of fast-moving electrons or positrons emitted from the nucleus.
There are two types of beta decay:
Beta Minus Decay
A neutron transforms into a proton, electron, and antineutrino.
[
n \rightarrow p + e^- + \bar{\nu}
]
Beta Plus Decay
A proton transforms into a neutron, positron, and neutrino.
[
p \rightarrow n + e^+ + \nu
]
Characteristics of beta radiation:
- Moderate penetration
- Medium ionization ability
- Can pass through paper but stopped by thin metal sheets.
Gamma Radiation



Gamma radiation consists of high-energy electromagnetic waves emitted from excited nuclei.
Gamma decay occurs when a nucleus transitions from a higher energy state to a lower energy state.
Characteristics:
- No mass
- No electric charge
- Very high penetration power
- Weak ionizing ability
Gamma rays require thick lead or concrete shielding.
Radioactive Decay Law
Radioactive decay follows an exponential law.
The rate of decay is proportional to the number of radioactive atoms present.
[
\frac{dN}{dt} = -\lambda N
]
where
- (N) = number of radioactive atoms
- (\lambda) = decay constant
The solution to this equation is
[
N = N_0 e^{-\lambda t}
]
Half-Life
The half-life of a radioactive substance is the time required for half of the radioactive nuclei to decay.
[
T_{1/2} = \frac{0.693}{\lambda}
]
Half-life is an important property used to measure the stability of radioactive isotopes.
Examples:
- Carbon-14: 5730 years
- Uranium-238: 4.5 billion years
Activity of Radioactive Substances
The activity of a radioactive sample measures the rate of decay.
[
A = \lambda N
]
Units of activity include:
- Becquerel (Bq): one decay per second
- Curie (Ci): (3.7 \times 10^{10}) decays per second
The curie unit was named after Marie Curie.
Radioactive Series
Many radioactive elements decay through a sequence of steps known as a radioactive decay series.
Examples include:
- Uranium series
- Thorium series
- Actinium series
Each series ends in a stable isotope.
Natural and Artificial Radioactivity
Radioactivity can be classified into two types.
Natural Radioactivity
Occurs naturally in elements such as:
- Uranium
- Thorium
- Radium
Artificial Radioactivity
Produced by nuclear reactions in laboratories.
Artificial radioactivity was discovered by Irène Joliot-Curie and Frédéric Joliot-Curie.
Detection of Radioactivity
Radioactive radiation can be detected using specialized instruments.
Common detectors include:
- Geiger–Müller counter
- Scintillation counter
- Cloud chamber
- Photographic plates
These instruments measure radiation intensity and particle properties.
Applications of Radioactivity
Radioactivity has many important applications.
Medicine
Radioactive isotopes are used for:
- Cancer treatment (radiotherapy)
- Medical imaging
- Diagnostic tests
Industry
Radioactive sources are used in:
- Thickness measurement
- Quality control
- Industrial radiography
Archaeology
Radiocarbon dating uses carbon-14 to determine the age of ancient artifacts.
Nuclear Energy
Nuclear reactors generate electricity using nuclear fission reactions involving radioactive materials.
Agriculture
Radioactive tracers help study plant growth and nutrient absorption.
Biological Effects of Radiation
Radiation can affect living organisms by damaging cells and DNA.
High radiation exposure can cause:
- Radiation sickness
- Genetic mutations
- Cancer
However, controlled radiation is beneficial in medical treatments.
Radiation Protection
To minimize radiation exposure, safety principles include:
- Limiting exposure time
- Increasing distance from sources
- Using shielding materials
These principles protect workers and patients from harmful radiation.
Radioactivity in the Environment
Radioactive materials exist naturally in the environment.
Sources include:
- Cosmic radiation
- Natural radioactive elements in soil
- Radon gas in the atmosphere
Background radiation is always present but usually harmless at low levels.
Radioactivity in Astrophysics
Radioactive decay processes occur in stars and supernova explosions.
These processes contribute to the formation of heavy elements in the universe.
Radioactive isotopes in meteorites provide information about the age of the solar system.
Modern Research in Radioactivity
Modern nuclear physics continues to explore radioactive phenomena.
Research areas include:
- Exotic nuclei
- Nuclear reactions
- Particle physics
- Radiation detection technology
Advances in this field contribute to medicine, energy production, and scientific discovery.
Conclusion
Radioactivity is a natural phenomenon in which unstable atomic nuclei emit radiation to achieve greater stability. The discovery of radioactivity revealed that atoms are not indivisible and that nuclear transformations occur within matter.
Radioactive decay involves the emission of alpha particles, beta particles, and gamma rays. These processes follow predictable laws and are characterized by quantities such as decay constant and half-life.
The study of radioactivity has had a profound impact on science and technology. It has led to advances in nuclear energy, medical treatments, industrial applications, and scientific research. Despite its potential hazards, radioactivity remains one of the most powerful tools for understanding the structure of matter and the processes occurring within atomic nuclei.
