
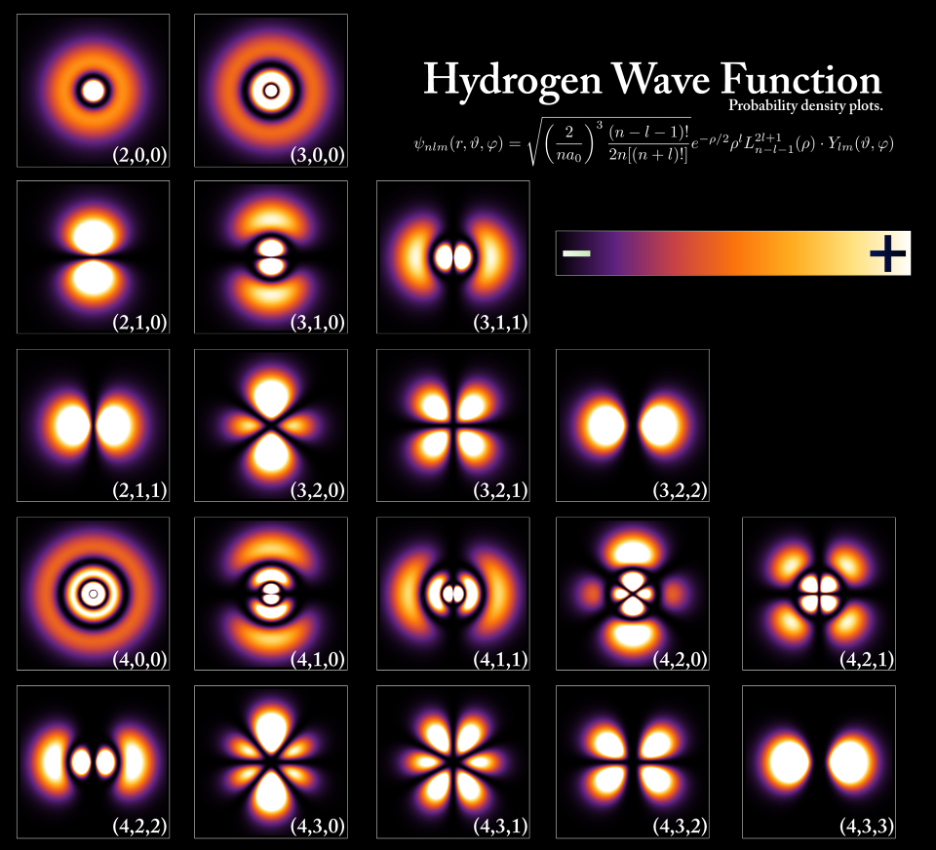

1. Introduction to Quantum Numbers
Quantum numbers are a set of numerical values used in quantum mechanics to describe the energy, position, orientation, and spin of electrons within an atom. They arise from the mathematical solutions of the Schrödinger wave equation and are fundamental to understanding the quantum mechanical model of the atom.
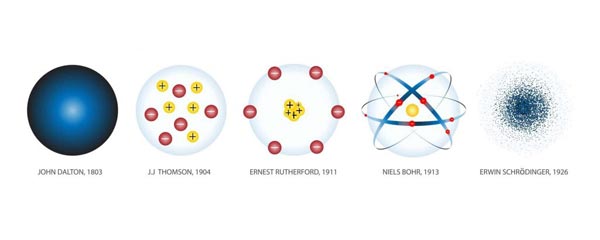
In classical atomic models, electrons were thought to move in fixed circular orbits around the nucleus. However, the development of quantum mechanics revealed that electrons behave both as particles and waves, making their exact location impossible to determine precisely. Instead, electrons exist in regions of probability called atomic orbitals.
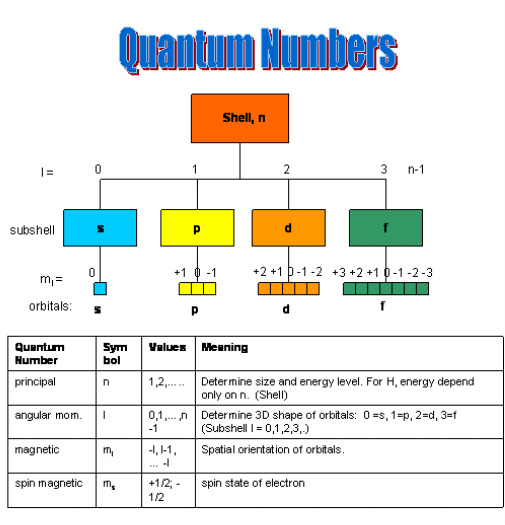
Quantum numbers help describe these orbitals and specify the quantum state of an electron. Each electron in an atom is characterized by a unique set of four quantum numbers:
- Principal quantum number (n)
- Azimuthal or angular momentum quantum number (l)
- Magnetic quantum number (mₗ)
- Spin quantum number (mₛ)
These numbers determine how electrons are arranged within atoms and explain many atomic properties such as electron configuration, chemical bonding, and periodic trends.
Quantum numbers are essential in modern chemistry, atomic physics, spectroscopy, and quantum mechanics. They provide a systematic way to describe the complex behavior of electrons within atoms and molecules.
2. Origin of Quantum Numbers




Quantum numbers originate from the development of quantum mechanics in the early twentieth century. Several important discoveries contributed to this development.
Wave–Particle Duality
Louis de Broglie proposed that particles such as electrons exhibit both wave-like and particle-like properties. This concept is known as wave–particle duality.
Electrons behave as waves when moving around the nucleus, leading to the formation of standing wave patterns that correspond to allowed energy levels.
Schrödinger Wave Equation
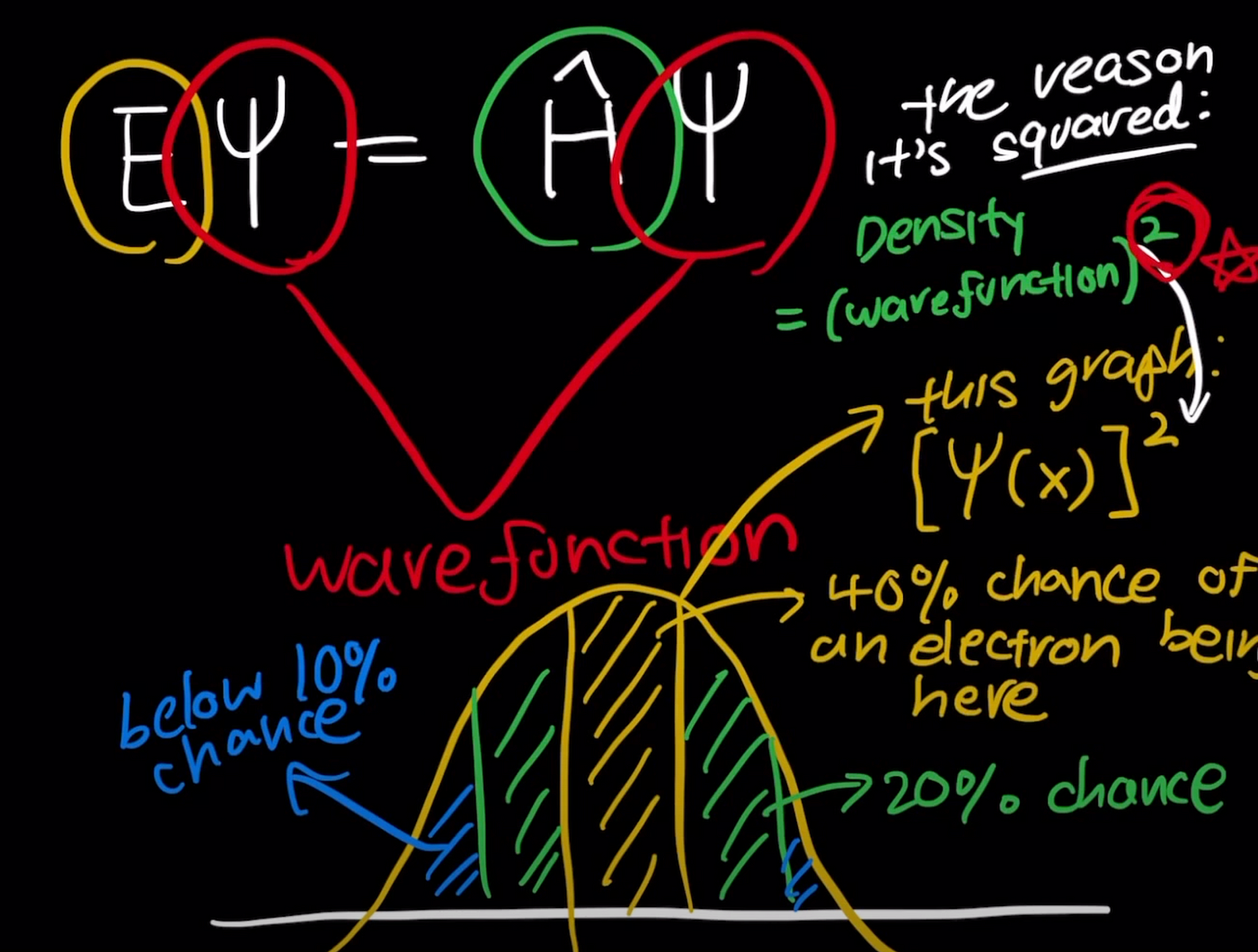
Erwin Schrödinger developed a mathematical equation describing how the quantum state of a physical system changes over time.
Solutions to this equation produce wave functions, which describe the probability of finding an electron in a particular region around the nucleus.
The solutions of the Schrödinger equation introduce quantum numbers that define these wave functions.
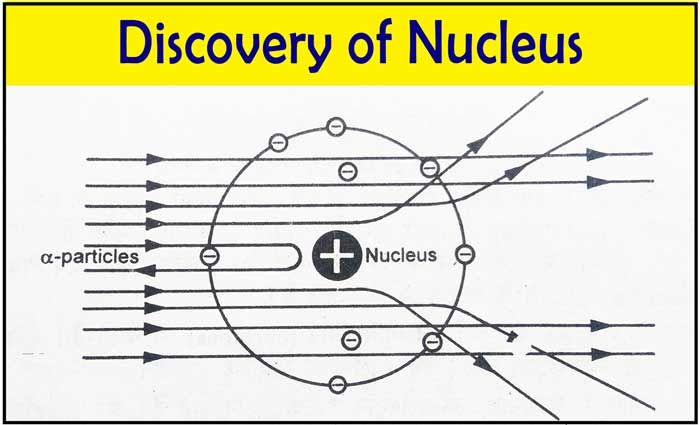
Heisenberg Uncertainty Principle
Werner Heisenberg showed that it is impossible to simultaneously determine the exact position and momentum of an electron.
Because of this uncertainty, electrons cannot be described as moving in fixed paths. Instead, their behavior is represented by probability distributions.
Quantum numbers therefore provide a way to describe the properties of these probability distributions.
3. Principal Quantum Number (n)




The principal quantum number, represented by the symbol n, describes the main energy level or shell of an electron within an atom.
Possible Values
The principal quantum number can take positive integer values:
n = 1, 2, 3, 4, …
Each value corresponds to a different electron shell.
Energy Levels
As the value of n increases:
- The energy of the electron increases.
- The average distance from the nucleus increases.
- The size of the orbital becomes larger.
Shell Names
Electron shells are often labeled using letters:
n = 1 → K shell
n = 2 → L shell
n = 3 → M shell
n = 4 → N shell
Maximum Number of Electrons
Each shell can hold a maximum number of electrons determined by:
Maximum electrons = 2n²
Examples:
n = 1 → 2 electrons
n = 2 → 8 electrons
n = 3 → 18 electrons
n = 4 → 32 electrons
The principal quantum number therefore determines the overall size and energy of orbitals.
4. Azimuthal Quantum Number (l)



The azimuthal quantum number, also called the angular momentum quantum number, is represented by l.
It describes the shape of the electron orbital.
Possible Values
The value of l depends on the principal quantum number n.
Possible values range from:
l = 0 to (n − 1)
Example:
If n = 3
Possible values of l are:
l = 0, 1, 2
Subshells
Each value of l corresponds to a specific subshell.
l = 0 → s subshell
l = 1 → p subshell
l = 2 → d subshell
l = 3 → f subshell
These subshells have different shapes and energy levels.
Orbital Shapes
- s orbitals are spherical.
- p orbitals have dumbbell shapes.
- d orbitals have cloverleaf shapes.
- f orbitals have complex shapes.
These shapes influence how atoms interact and bond with other atoms.
5. Magnetic Quantum Number (mₗ)



The magnetic quantum number, represented by mₗ, describes the orientation of an orbital in space.
Possible Values
The value of mₗ ranges from:
−l to +l
including zero.
Example:
If l = 1
Possible mₗ values:
−1, 0, +1
This means that the p subshell contains three orbitals.
Number of Orbitals
The number of orbitals in a subshell can be calculated using:
2l + 1
Examples:
s subshell (l = 0) → 1 orbital
p subshell (l = 1) → 3 orbitals
d subshell (l = 2) → 5 orbitals
f subshell (l = 3) → 7 orbitals
Each orbital can hold up to two electrons.
6. Spin Quantum Number (mₛ)


The spin quantum number, represented by mₛ, describes the spin direction of an electron.
Electrons behave as if they rotate around their own axis, producing a magnetic field.
Possible Values
The spin quantum number can have only two values:
+1/2
−1/2
These represent two possible spin orientations.
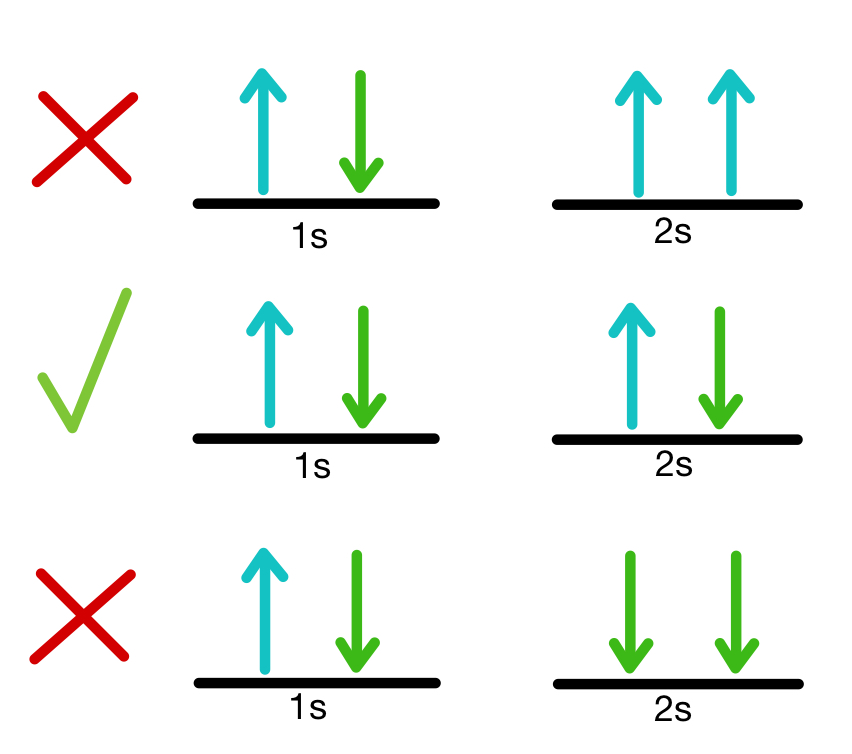
Pauli Exclusion Principle
The Pauli Exclusion Principle states that:
No two electrons in the same atom can have the same set of four quantum numbers.
Because of this principle:
- Each orbital can hold a maximum of two electrons.
- The electrons must have opposite spins.
This rule is fundamental for determining electron configurations.
7. Relationship Between Quantum Numbers and Atomic Orbitals
Quantum numbers define the characteristics of atomic orbitals.
Each orbital is uniquely defined by three quantum numbers:
n, l, and mₗ.
Electrons occupying these orbitals are further distinguished by the spin quantum number.
Example
Consider the orbital:
n = 2
l = 1
This corresponds to the 2p subshell.
Possible mₗ values:
−1, 0, +1
Therefore, the 2p subshell contains three orbitals.
Each orbital can contain two electrons, giving a maximum of six electrons in the 2p subshell.
Quantum numbers therefore determine the entire electronic structure of atoms.
8. Quantum Numbers and Electron Configuration
Quantum numbers help explain how electrons fill orbitals in atoms.
Three important principles guide electron arrangement.
Aufbau Principle
Electrons occupy orbitals with the lowest energy first.
Pauli Exclusion Principle
Each orbital holds a maximum of two electrons with opposite spins.
Hund’s Rule
Electrons occupy orbitals singly before pairing.
These principles explain the electron configurations of elements in the periodic table.
9. Quantum Numbers and the Periodic Table




The structure of the periodic table is closely related to quantum numbers.
s-block
Elements in the first two groups fill the s subshell.
p-block
Elements in groups 13–18 fill p orbitals.
d-block
Transition metals involve filling of d orbitals.
f-block
Lanthanides and actinides involve f orbitals.
Quantum numbers therefore explain the arrangement of elements in the periodic table.
10. Importance of Quantum Numbers
Quantum numbers are crucial for understanding many aspects of chemistry and physics.
Atomic Structure
They describe the arrangement of electrons within atoms.
Chemical Bonding
Orbital shapes and orientations determine bonding patterns.
Spectroscopy
Quantum transitions between energy levels produce spectral lines.
Magnetism
Electron spin explains magnetic properties of materials.
Quantum Chemistry
Quantum numbers form the basis for calculations of molecular structures and reactions.
11. Applications of Quantum Numbers
Quantum numbers have numerous applications in modern science and technology.
Semiconductor Technology
Understanding electron orbitals is essential for designing electronic devices.
Laser Technology
Laser operation relies on electron transitions between quantum energy levels.
Magnetic Resonance Imaging (MRI)
MRI uses electron and nuclear spin properties.
Spectroscopy
Quantum numbers help identify elements and compounds through spectral analysis.
Nanotechnology
Quantum behavior becomes significant at very small scales.
12. Conclusion
Quantum numbers provide a powerful framework for describing the behavior of electrons within atoms. By defining the energy, shape, orientation, and spin of electron orbitals, quantum numbers form the foundation of modern atomic theory.
The four quantum numbers—principal, azimuthal, magnetic, and spin—collectively describe the quantum state of electrons. These numbers explain the structure of atomic orbitals, electron configurations, and the organization of the periodic table.
Understanding quantum numbers is essential for studying advanced topics in chemistry, physics, and materials science. They enable scientists to predict atomic properties, chemical bonding patterns, and the behavior of matter at the microscopic level.
As research in quantum mechanics continues to evolve, quantum numbers remain a fundamental concept in understanding the complex and fascinating world of atoms and subatomic particles.