



Introduction
Semiconductors are materials whose electrical conductivity lies between that of conductors and insulators. Unlike metals, which conduct electricity very well, and insulators, which hardly conduct electricity at all, semiconductors have conductivity that can be controlled and modified by factors such as temperature, impurities, electric fields, and light.
Semiconductors form the foundation of modern electronics. Almost every electronic device—computers, smartphones, televisions, solar cells, and communication systems—depends on semiconductor technology.
The behavior of semiconductors is explained using band theory, which describes how electrons move through energy bands in solids. In semiconductors, a small energy gap exists between the valence band and the conduction band, allowing electrons to move to the conduction band under certain conditions.
Common semiconductor materials include:
- Silicon (Si)
- Germanium (Ge)
- Gallium arsenide (GaAs)
These materials are widely used to manufacture electronic components such as diodes, transistors, and integrated circuits.
The development of semiconductor technology led to the invention of the transistor by John Bardeen, Walter Brattain, and William Shockley.
Basic Properties of Semiconductors
Semiconductors exhibit several important electrical properties.
Moderate Electrical Conductivity
Their conductivity lies between conductors and insulators.
Temperature Dependence
Electrical conductivity increases as temperature rises.
Controlled Conductivity
Adding impurities (doping) can dramatically change electrical behavior.
Charge Carriers
Electrical conduction occurs through electrons and holes.
These properties make semiconductors highly useful for electronic applications.
Crystal Structure of Semiconductors
Most semiconductor materials have crystalline structures where atoms are arranged in a regular pattern.
For example, silicon atoms form a diamond cubic crystal structure.
Each silicon atom shares electrons with four neighboring atoms through covalent bonds.
This bonding structure creates a stable lattice that determines the electronic properties of the semiconductor.
Energy Band Structure


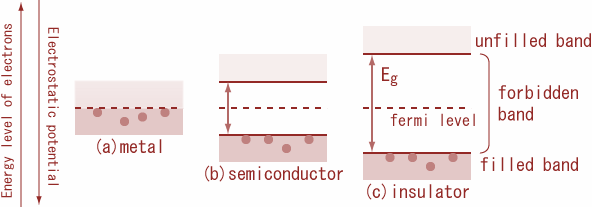
The electronic properties of semiconductors can be explained using energy band theory.
In solids, electrons occupy energy bands rather than discrete energy levels.
The main bands include:
Valence Band
The valence band contains electrons involved in chemical bonding.
Conduction Band
The conduction band contains free electrons that can move through the material and conduct electricity.
Band Gap
The energy difference between the valence band and conduction band is called the band gap.
For semiconductors, this gap is relatively small.
Typical band gaps:
- Silicon: about 1.1 eV
- Germanium: about 0.7 eV
Because the band gap is small, electrons can be excited into the conduction band with modest energy input.
Intrinsic Semiconductors




An intrinsic semiconductor is a pure semiconductor without any added impurities.
At absolute zero temperature:
- All electrons remain in the valence band.
- The material behaves like an insulator.
As temperature increases:
- Some electrons gain enough energy to jump to the conduction band.
- Each excited electron leaves behind a hole in the valence band.
Thus, conduction occurs due to:
- Electrons in the conduction band
- Holes in the valence band
In intrinsic semiconductors, the number of electrons equals the number of holes.
Electron–Hole Pairs
When electrons move from the valence band to the conduction band, they leave behind empty states known as holes.
A hole behaves like a positively charged particle.
Both electrons and holes contribute to electrical conduction.
The generation of electron–hole pairs occurs due to:
- Thermal energy
- Light absorption
- Electrical excitation
Extrinsic Semiconductors


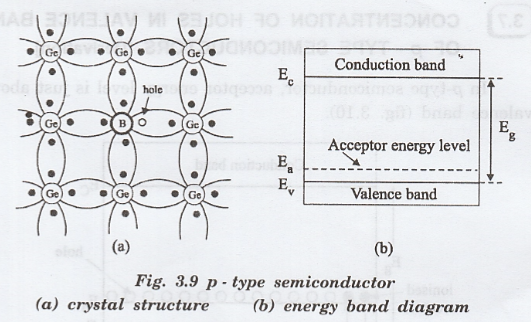
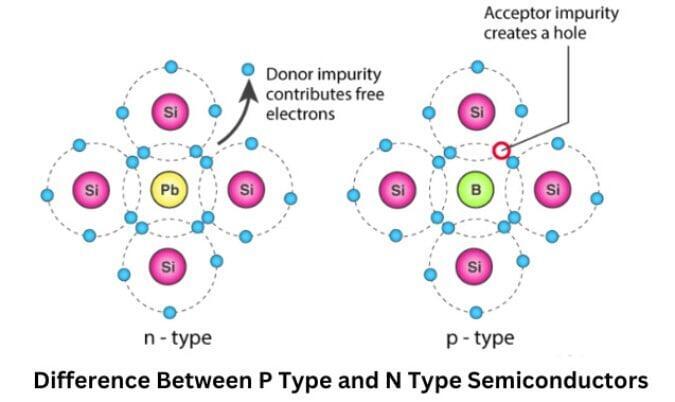
Extrinsic semiconductors are formed by adding small amounts of impurities to intrinsic semiconductors.
This process is called doping.
Doping greatly increases electrical conductivity.
Two main types exist.
n-Type Semiconductors
In n-type semiconductors, impurities with five valence electrons are added.
Examples include:
- Phosphorus
- Arsenic
- Antimony
These atoms donate extra electrons to the conduction band.
As a result:
- Electrons become the majority carriers
- Holes become minority carriers
p-Type Semiconductors
In p-type semiconductors, impurities with three valence electrons are added.
Examples include:
- Boron
- Aluminum
- Gallium
These atoms create holes in the valence band.
Thus:
- Holes become majority carriers
- Electrons become minority carriers
Fermi Energy Level
The Fermi level represents the highest occupied energy level at absolute zero temperature.
In intrinsic semiconductors:
- The Fermi level lies near the center of the band gap.
In n-type semiconductors:
- The Fermi level shifts closer to the conduction band.
In p-type semiconductors:
- The Fermi level shifts toward the valence band.
The position of the Fermi level determines the electrical behavior of the semiconductor.
p-n Junction



A p-n junction is formed when p-type and n-type semiconductor materials are joined together.
At the junction:
- Electrons from the n-type region diffuse into the p-type region.
- Holes move in the opposite direction.
This diffusion creates a depletion region where mobile charge carriers are absent.
The depletion region acts as a barrier that controls the flow of current.
Semiconductor Diodes
A diode is an electronic device formed from a p-n junction.
It allows current to flow in one direction but blocks it in the opposite direction.
Types of semiconductor diodes include:
- Rectifier diodes
- Zener diodes
- Light-emitting diodes (LEDs)
- Photodiodes
Transistors
Transistors are semiconductor devices used for amplification and switching.
Two main types exist:
Bipolar Junction Transistor (BJT)
Consists of three layers of semiconductor material.
Field Effect Transistor (FET)
Uses electric fields to control current flow.
Transistors are the fundamental building blocks of modern electronic circuits.
Semiconductor Applications
Semiconductors are essential in many technologies.
Integrated Circuits
Microchips used in computers contain billions of transistors made from semiconductors.
Solar Cells
Photovoltaic cells convert sunlight into electricity using semiconductor materials.
LEDs
Light-emitting diodes produce light through electron-hole recombination.
Sensors
Semiconductors are used in temperature, light, and pressure sensors.
Communication Technology
Semiconductors enable modern communication systems such as mobile networks and satellite technology.
Semiconductor Materials
Several materials are commonly used in semiconductor devices.
Silicon
Most widely used semiconductor.
Germanium
Used in early electronic devices.
Gallium Arsenide
Used in high-speed and optoelectronic devices.
Silicon Carbide
Used in high-power electronics.
Each material has unique electrical properties.
Modern Semiconductor Research
Research continues to explore new semiconductor materials and technologies.
Examples include:
- Nanotechnology
- Two-dimensional materials
- Quantum semiconductor devices
These advances aim to create faster, smaller, and more efficient electronic devices.
Conclusion
Semiconductors are materials with electrical conductivity between conductors and insulators. Their conductivity can be precisely controlled by temperature, impurities, and external fields, making them ideal for electronic applications.
The behavior of semiconductors is explained using band theory, which describes how electrons move between the valence and conduction bands. Through doping, intrinsic semiconductors can be transformed into n-type and p-type materials with enhanced electrical conductivity.
Semiconductor devices such as diodes and transistors form the foundation of modern electronics. These components enable the operation of computers, communication systems, and many other technologies. As research continues, semiconductor science remains one of the most important fields driving technological innovation in the modern world.
