
Introduction
Gibbs Free Energy is one of the most important concepts in thermodynamics and physical chemistry. It is used to determine whether a chemical reaction or physical process will occur spontaneously under constant temperature and pressure. Named after the American scientist Josiah Willard Gibbs, this thermodynamic potential combines the effects of enthalpy (heat content) and entropy (degree of disorder) to predict the direction of chemical reactions.
In many natural and industrial processes, systems tend to move toward states that minimize free energy. Gibbs Free Energy provides a quantitative way to understand this tendency and to predict the feasibility of reactions.
The concept of Gibbs Free Energy is widely used in many areas of science, including:
- Chemistry
- Biology
- Materials science
- Environmental science
- Chemical engineering
It is particularly important in understanding chemical equilibrium, biochemical reactions, and energy transformations in living organisms.
1. Definition of Gibbs Free Energy
Gibbs Free Energy represents the maximum amount of useful work that can be obtained from a thermodynamic system at constant temperature and pressure.
It is defined mathematically as:
G = H – TS
Where:
G = Gibbs Free Energy
H = Enthalpy
T = Absolute temperature (Kelvin)
S = Entropy
This equation shows that free energy depends on both the heat content of the system and the disorder within the system.
2. Change in Gibbs Free Energy
The change in Gibbs Free Energy during a chemical reaction is expressed as:
\Delta G = \Delta H – T\Delta S
Where:
ΔG = change in Gibbs Free Energy
ΔH = change in enthalpy
T = absolute temperature
ΔS = change in entropy
This equation is fundamental in predicting whether a reaction will occur spontaneously.
3. Interpretation of Gibbs Free Energy
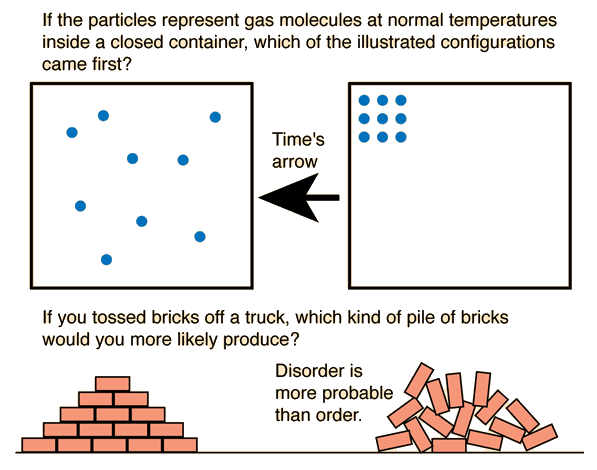
The sign of ΔG determines the spontaneity of a process.
Negative ΔG
If ΔG is negative:
- The process is spontaneous
- The reaction can occur without external energy input
Examples include combustion reactions and many biochemical reactions.
Positive ΔG
If ΔG is positive:
- The reaction is non-spontaneous
- External energy is required for the reaction to occur
An example is photosynthesis, which requires energy from sunlight.
ΔG Equal to Zero
If ΔG equals zero:
- The system is in equilibrium
- The forward and reverse reactions occur at the same rate
At equilibrium, no net change occurs in the system.
4. Relationship Between Enthalpy, Entropy, and Gibbs Free Energy

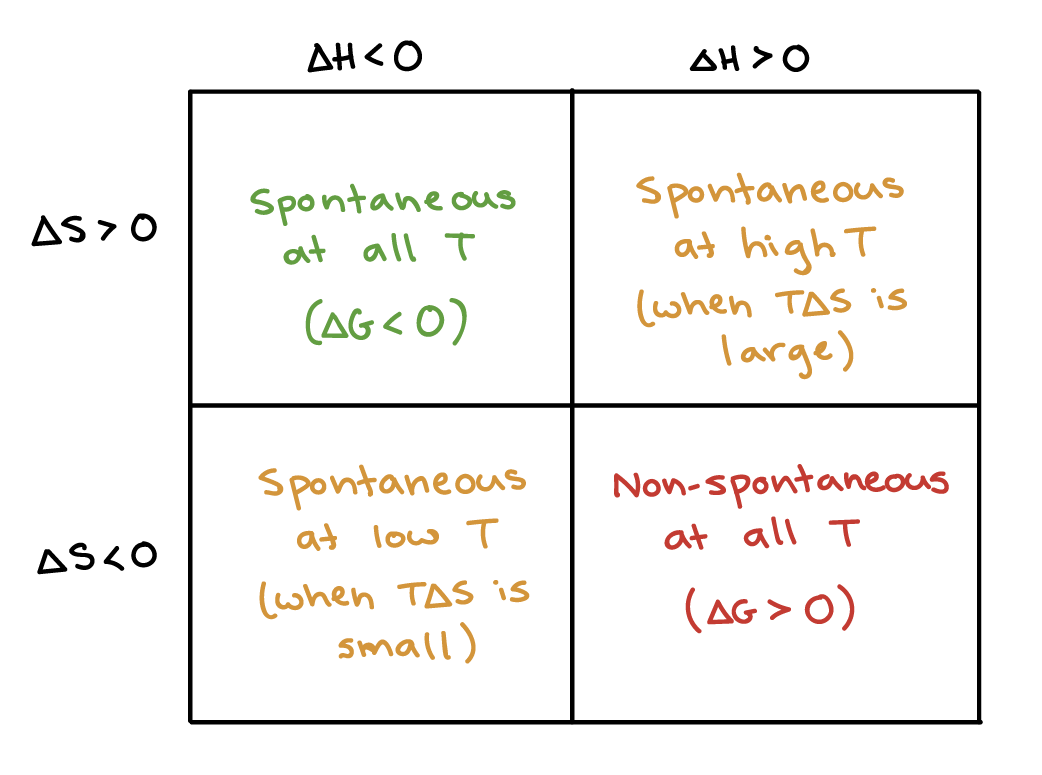

The Gibbs Free Energy equation shows how enthalpy and entropy influence reaction spontaneity.
There are four possible combinations of enthalpy and entropy changes.
Case 1: ΔH Negative, ΔS Positive
- Reaction releases heat
- Disorder increases
These reactions are always spontaneous.
Example:
Combustion reactions.
Case 2: ΔH Positive, ΔS Negative
- Heat absorbed
- Disorder decreases
These reactions are never spontaneous.
Case 3: ΔH Negative, ΔS Negative
Reaction may be spontaneous at low temperatures.
Case 4: ΔH Positive, ΔS Positive
Reaction may be spontaneous at high temperatures.
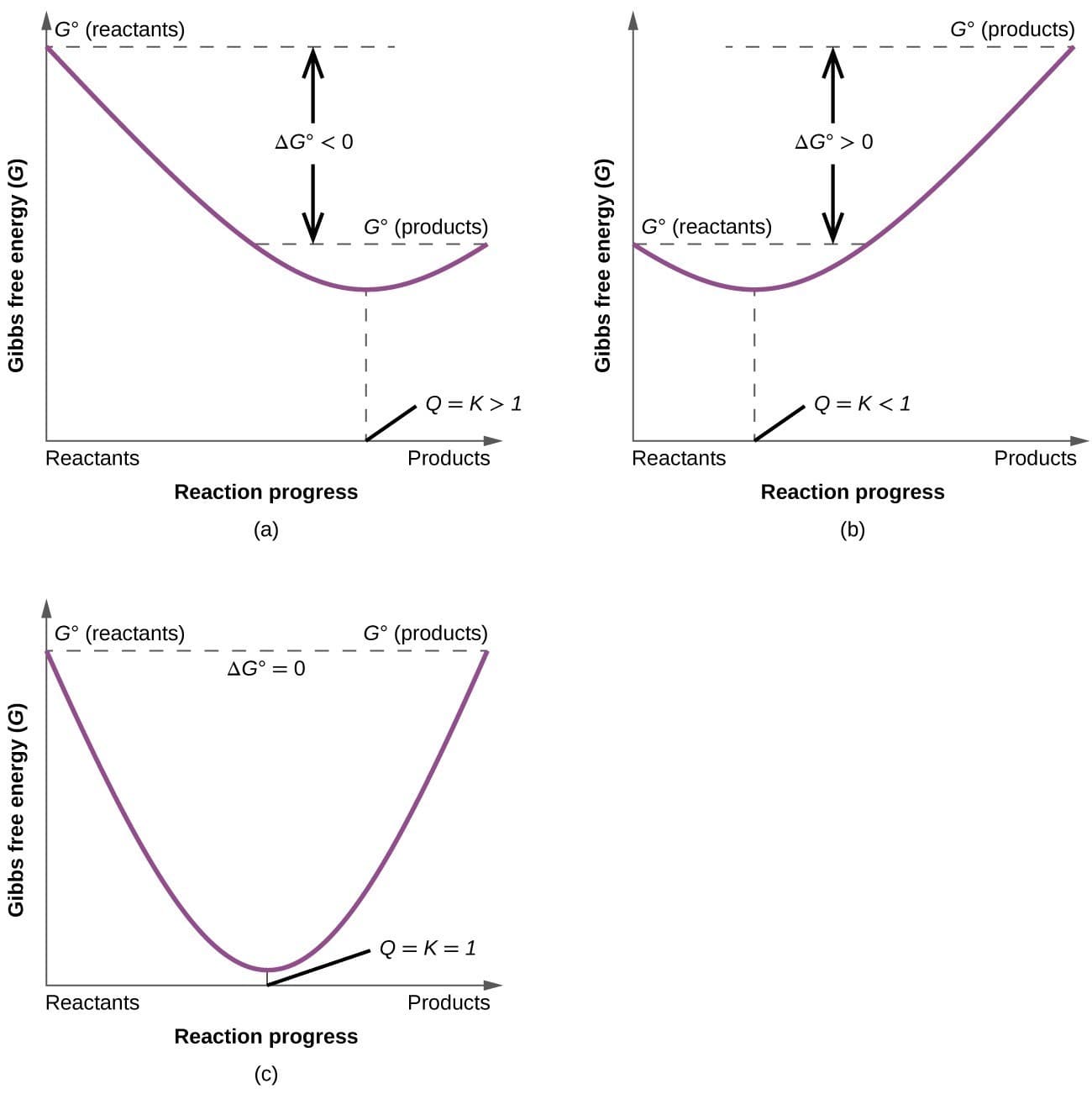
5. Free Energy and Chemical Equilibrium
Gibbs Free Energy is closely related to chemical equilibrium.
At equilibrium:
ΔG = 0
The relationship between free energy and the equilibrium constant is given by:
\Delta G^{\circ} = -RT \ln K
Where:
ΔG° = standard free energy change
R = gas constant
T = temperature in Kelvin
K = equilibrium constant
Interpretation
If K > 1 → reaction favors products
If K < 1 → reaction favors reactants
This relationship allows chemists to predict equilibrium conditions.
6. Standard Gibbs Free Energy
Standard Gibbs Free Energy refers to free energy change measured under standard conditions.
Standard conditions include:
- Temperature = 298 K
- Pressure = 1 atm
- Concentration = 1 mol/L
Standard values are used for comparing reactions and calculating thermodynamic properties.
7. Gibbs Free Energy in Chemical Reactions
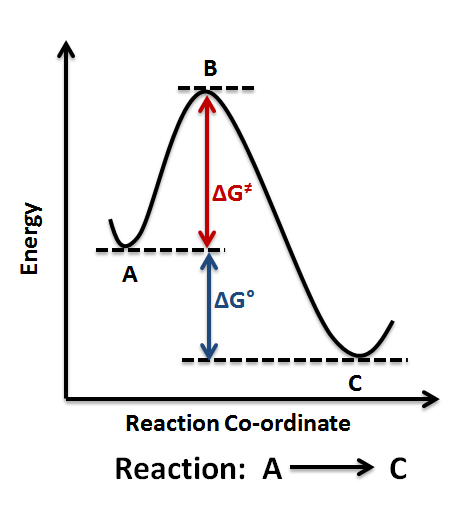



Chemical reactions can be classified based on free energy changes.
Exergonic Reactions
Exergonic reactions release free energy.
Characteristics:
- ΔG is negative
- Reaction occurs spontaneously
Example:
Cellular respiration.
Endergonic Reactions
Endergonic reactions require energy input.
Characteristics:
- ΔG is positive
- Reaction does not occur spontaneously
Example:
Photosynthesis.
8. Temperature Dependence of Gibbs Free Energy
Temperature plays a significant role in determining reaction spontaneity.
From the equation:
ΔG = ΔH − TΔS
Increasing temperature increases the influence of entropy.
Some reactions become spontaneous only at high temperatures.
Example:
Melting of ice occurs when temperature rises above 0°C.
9. Gibbs Free Energy in Biological Systems
Gibbs Free Energy is essential in understanding biochemical reactions.
Living organisms rely on energy transformations to sustain life.
ATP and Free Energy
The molecule ATP (adenosine triphosphate) stores and releases energy for cellular processes.
Hydrolysis of ATP releases free energy used in:
- Muscle contraction
- Protein synthesis
- Active transport
Metabolism
Metabolic pathways involve sequences of reactions that either release or consume free energy.
Cells maintain energy balance through these processes.
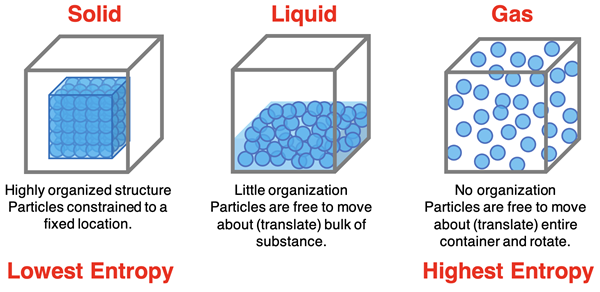
10. Gibbs Free Energy and Phase Transitions
Gibbs Free Energy also explains phase changes such as melting, freezing, and vaporization.
At equilibrium between two phases:
Gsolid = Gliquid
or
Gliquid = Ggas
Phase transitions occur when free energy of one phase becomes lower than another.
11. Applications of Gibbs Free Energy
Gibbs Free Energy is widely used across many scientific and engineering disciplines.
Chemical Engineering
Engineers use free energy calculations to design chemical reactors and optimize industrial processes.
Electrochemistry
Free energy changes determine electrical energy produced by batteries and fuel cells.
Environmental Science
Free energy helps explain chemical reactions occurring in the atmosphere and oceans.
Materials Science
Free energy analysis helps determine the stability of materials and phase transformations.
12. Importance of Gibbs Free Energy
Gibbs Free Energy is a powerful concept that integrates multiple thermodynamic principles into a single framework. By combining enthalpy and entropy, it allows scientists to predict the direction and feasibility of reactions.
The concept helps explain why some reactions occur naturally while others require energy input. It also provides insights into equilibrium conditions, reaction mechanisms, and energy efficiency.
Understanding Gibbs Free Energy is essential for studying chemical reactions, biological systems, and energy transformations.
Conclusion
Gibbs Free Energy is one of the most important thermodynamic quantities used to determine whether chemical reactions and physical processes occur spontaneously. It combines the effects of enthalpy and entropy into a single equation that describes the usable energy available in a system.
A negative change in Gibbs Free Energy indicates a spontaneous process, while a positive change indicates that energy must be supplied. When free energy change is zero, the system is at equilibrium.
The Gibbs Free Energy equation also reveals how temperature influences reaction spontaneity and how energy transformations occur in chemical and biological systems.
From predicting chemical equilibrium to understanding metabolic reactions in living organisms, Gibbs Free Energy plays a critical role in modern science and technology. Its applications extend across chemistry, biology, engineering, and environmental science, making it one of the most fundamental concepts in thermodynamics.