1. Introduction to Nuclear Chemistry
Nuclear chemistry is the branch of chemistry that studies changes occurring in the atomic nucleus, including processes such as radioactive decay, nuclear reactions, nuclear fission, and nuclear fusion. Unlike ordinary chemical reactions, which involve the rearrangement of electrons in atoms or molecules, nuclear reactions involve changes in the nucleus of atoms, resulting in the formation of different elements or isotopes.
The nucleus of an atom contains protons and neutrons, collectively called nucleons. Nuclear chemistry focuses on the behavior, stability, and transformation of these nucleons.
Nuclear processes release enormous amounts of energy because the forces holding nucleons together are extremely strong. These energies are millions of times greater than those involved in typical chemical reactions.
Nuclear chemistry plays a vital role in many scientific and technological fields, including:
- Nuclear power generation
- Medical imaging and cancer treatment
- Archaeological dating techniques
- Environmental monitoring
- Space exploration
- Industrial applications
Understanding nuclear chemistry allows scientists to harness nuclear energy safely and apply nuclear techniques in medicine, agriculture, and scientific research.
2. Structure of the Atomic Nucleus
The atomic nucleus is the central part of an atom and contains most of its mass.
The nucleus consists of:
- Protons (positively charged particles)
- Neutrons (neutral particles)
Electrons move around the nucleus in orbitals, but they are not involved directly in nuclear reactions.
Nuclear Forces
Protons in the nucleus repel each other because they carry positive charges. However, they remain bound together due to the strong nuclear force, which is a powerful attractive force acting between nucleons at very short distances.
The stability of the nucleus depends on the balance between:
- Nuclear attraction
- Electrostatic repulsion
3. Isotopes




Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons.
Example:
Hydrogen has three isotopes:
- Protium (¹H)
- Deuterium (²H)
- Tritium (³H)
All isotopes have identical chemical properties because they have the same number of electrons. However, their nuclear properties may differ significantly.
Some isotopes are stable, while others are radioactive.
4. Radioactivity
Radioactivity is the spontaneous emission of radiation from unstable atomic nuclei.
Radioactive decay occurs when an unstable nucleus transforms into a more stable nucleus by emitting particles or energy.
Radioactive substances emit three main types of radiation:
- Alpha radiation
- Beta radiation
- Gamma radiation
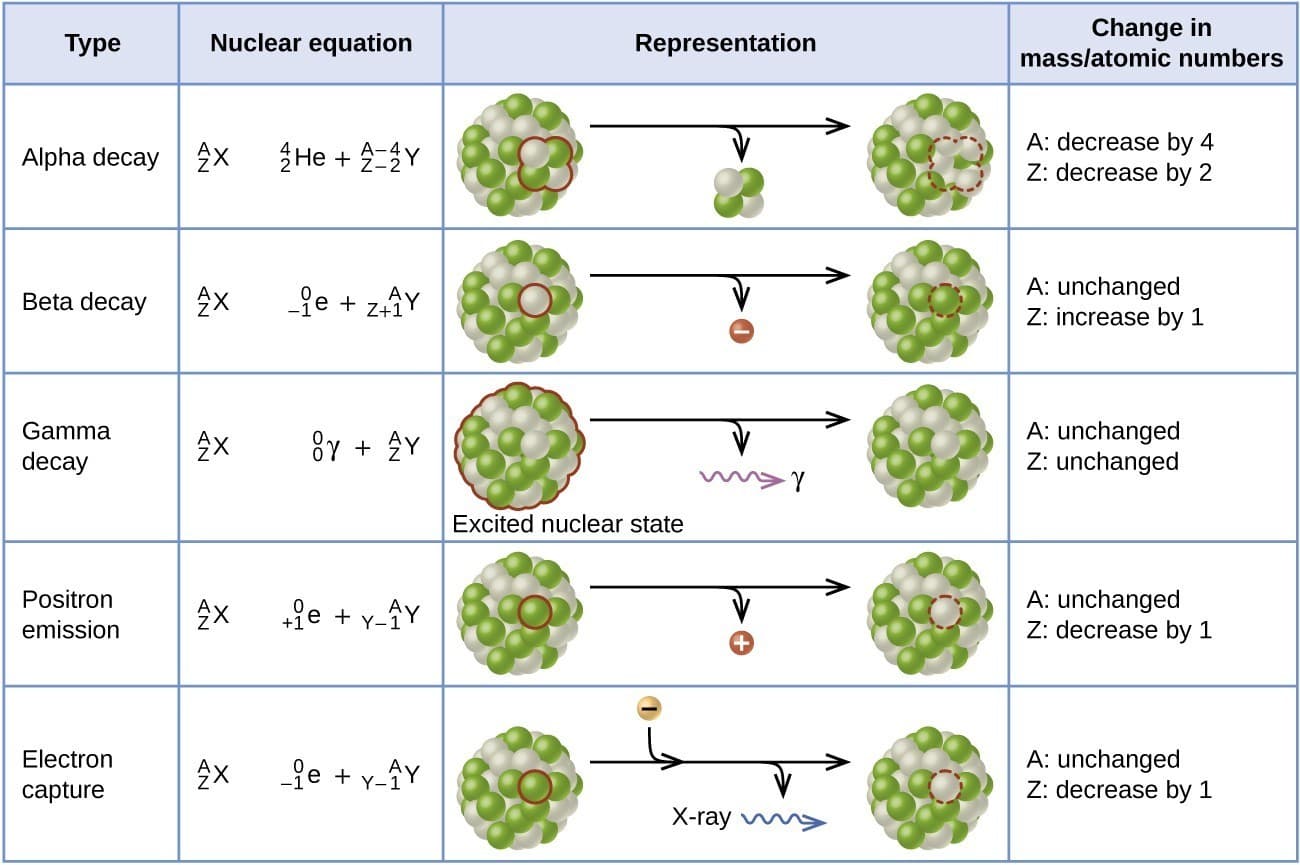
5. Types of Radioactive Radiation




Alpha Radiation (α)
Alpha particles consist of two protons and two neutrons.
Characteristics:
- Low penetration power
- High ionization power
- Can be stopped by paper
Example decay:
²³⁸U → ²³⁴Th + α
Beta Radiation (β)
Beta particles are high-speed electrons emitted from the nucleus.
Characteristics:
- Moderate penetration power
- Higher penetration than alpha particles
Example:
¹⁴C → ¹⁴N + β⁻
Gamma Radiation (γ)
Gamma rays are high-energy electromagnetic radiation.
Characteristics:
- Very high penetration power
- No mass and no charge
Gamma radiation usually accompanies alpha or beta decay.
6. Radioactive Decay Law
Radioactive decay follows first-order kinetics.
The rate of decay depends on the number of radioactive atoms present.
The decay equation is:
[
N = N_0 e^{- \lambda t}
]
Where:
- N = number of atoms remaining
- N₀ = initial number of atoms
- λ = decay constant
- t = time
7. Half-Life of Radioactive Elements
Half-life is the time required for half of the radioactive nuclei to decay.
Example:
Carbon-14 has a half-life of about 5730 years.
After one half-life:
50% of the original material remains.
After two half-lives:
25% remains.
Half-life is constant for each radioactive isotope.
8. Nuclear Stability

The stability of a nucleus depends on the neutron-to-proton ratio (N/Z ratio).
Light elements:
Stable when N ≈ Z.
Heavy elements:
Require more neutrons than protons for stability.
The band of stability is a region where stable isotopes exist.
Nuclei outside this band tend to undergo radioactive decay.
9. Nuclear Reactions
Nuclear reactions involve changes in the nucleus and result in the formation of new elements.
Example:
¹⁴N + α → ¹⁷O + p
Types of nuclear reactions include:
- Nuclear fission
- Nuclear fusion
- Artificial transmutation
10. Nuclear Fission




Nuclear fission is the process in which a heavy atomic nucleus splits into smaller nuclei, releasing a large amount of energy.
Example:
²³⁵U + n → ¹⁴¹Ba + ⁹²Kr + 3n + energy
Fission produces:
- Energy
- Neutrons
- Radioactive products
The emitted neutrons can trigger further reactions, leading to a chain reaction.
11. Nuclear Fusion




Nuclear fusion is the process in which light nuclei combine to form a heavier nucleus, releasing energy.
Example:
²H + ³H → ⁴He + n + energy
Fusion occurs in the Sun and stars, where extremely high temperatures allow nuclei to overcome electrostatic repulsion.
Fusion releases enormous energy and is considered a promising future energy source.
12. Nuclear Reactors
Nuclear reactors are devices used to control nuclear fission reactions.
Components of a reactor include:
- Fuel rods (uranium or plutonium)
- Moderator (slows neutrons)
- Control rods (absorb neutrons)
- Coolant (removes heat)
Reactors produce heat that is used to generate electricity.
13. Nuclear Energy
Nuclear energy is generated through fission or fusion reactions.
Advantages include:
- High energy output
- Low greenhouse gas emissions
- Reliable energy supply
However, challenges include:
- Radioactive waste management
- Nuclear accidents
- High construction costs
14. Applications of Nuclear Chemistry
Nuclear chemistry has many practical applications.
Medicine
- Cancer treatment (radiotherapy)
- Medical imaging (PET scans)
- Diagnostic tracers
Agriculture
- Crop improvement
- Pest control
- Food irradiation
Industry
- Thickness measurement
- Leak detection
- Sterilization processes
Archaeology
Radioactive dating techniques determine the age of ancient artifacts.
Example:
Carbon-14 dating.
15. Environmental and Safety Considerations
Radioactive materials must be handled carefully.
Radiation protection measures include:
- Shielding
- Monitoring exposure
- Safe storage of nuclear waste
Proper management ensures minimal environmental impact.
16. Importance of Nuclear Chemistry
Nuclear chemistry has revolutionized science and technology.
It helps scientists:
- Understand atomic structure
- Develop energy technologies
- Diagnose and treat diseases
- Study Earth’s history
- Explore space
Conclusion
Nuclear chemistry studies processes involving changes in the atomic nucleus, including radioactive decay, nuclear reactions, fission, and fusion. These processes release enormous energy and have numerous scientific and technological applications. Nuclear chemistry plays a critical role in energy production, medicine, environmental science, and industrial applications. By understanding nuclear processes and managing them safely, scientists can harness nuclear energy and technologies to benefit society while minimizing risks to humans and the environment.