

1. Introduction to Chemical Equations
Chemical equations are symbolic representations of chemical reactions. They show how substances known as reactants transform into new substances called products during a chemical reaction. Chemical equations provide a concise way to describe chemical changes, allowing scientists to understand what substances participate in a reaction and what products are formed.
A chemical equation uses chemical formulas, symbols, and coefficients to represent chemical reactions. Each equation communicates important information about the reaction, including the identity of substances involved, the proportions in which they react, and the physical states of the substances.
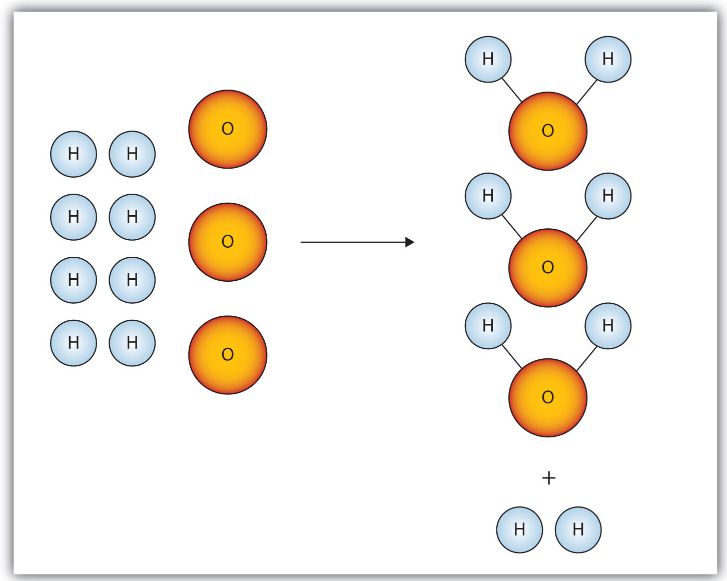
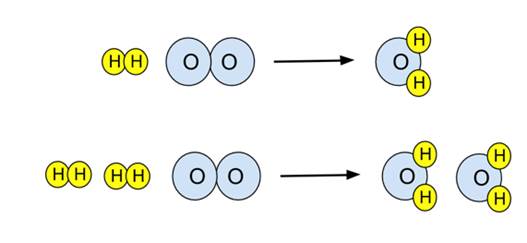
For example, the reaction between hydrogen and oxygen to form water can be represented as:
2H₂ + O₂ → 2H₂O
In this equation:
- Hydrogen and oxygen are reactants.
- Water is the product.
- The arrow indicates the direction of the reaction.
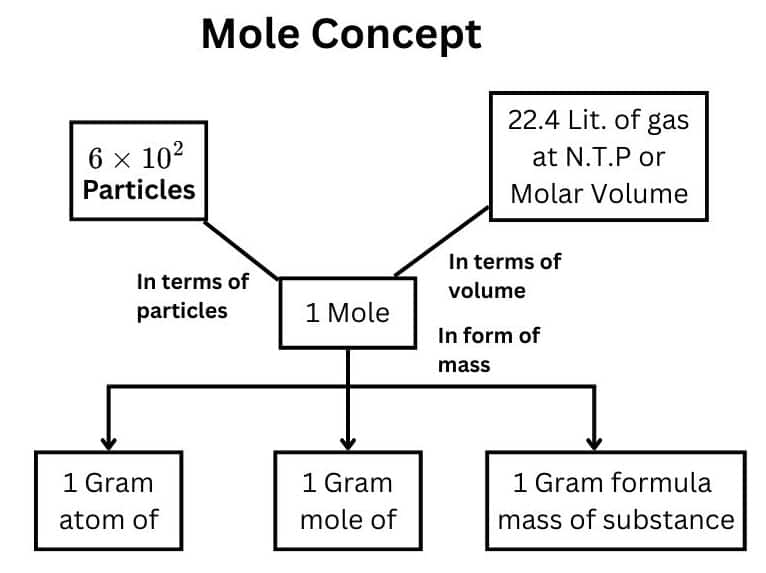
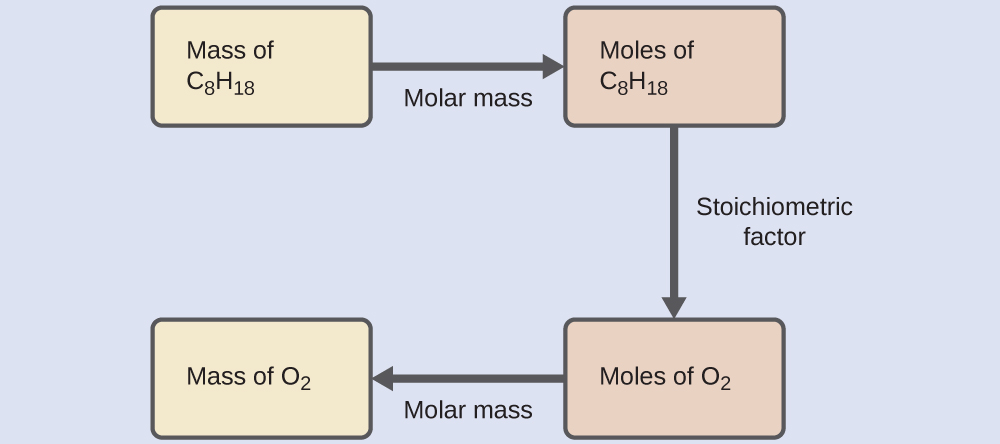
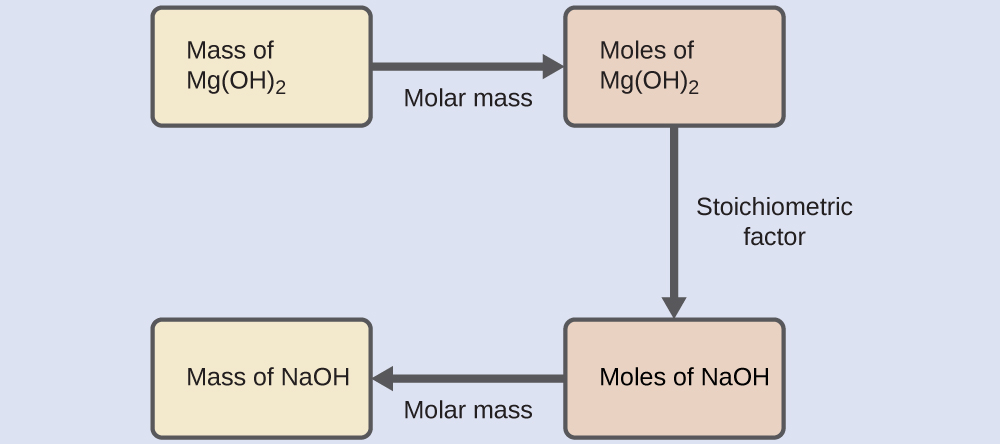
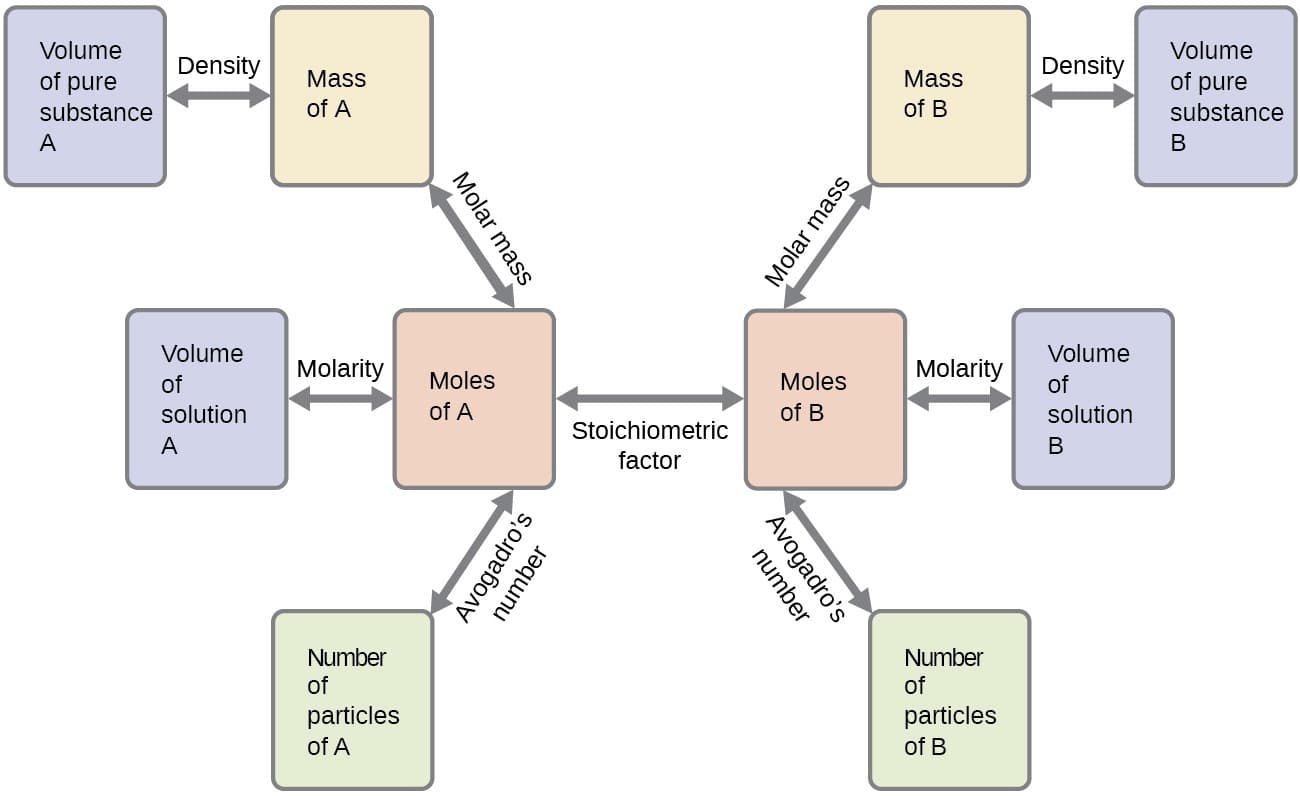
Chemical equations play a vital role in chemistry because they provide the basis for quantitative analysis of chemical reactions. By examining a chemical equation, chemists can determine the amounts of reactants required and the quantities of products formed.
Chemical equations are essential for studying many areas of chemistry, including:
- Stoichiometry
- Reaction mechanisms
- Chemical kinetics
- Thermodynamics
- Industrial chemical processes
Understanding chemical equations allows scientists to predict reaction outcomes and design chemical processes efficiently.
2. Components of Chemical Equations




A chemical equation consists of several key components that together represent a chemical reaction.
Reactants
Reactants are the starting substances that undergo chemical change during a reaction. They appear on the left side of the equation.
Example:
H₂ + O₂
These substances interact with each other to produce new substances.
Products
Products are the substances formed as a result of the chemical reaction. They appear on the right side of the equation.
Example:
H₂O
Products may have properties that differ significantly from those of the reactants.
Arrow Symbol
The arrow symbol (→) represents the direction of the reaction. It indicates that reactants are transformed into products.
Plus Sign
The plus sign (+) separates multiple reactants or products in a chemical equation.
Example:
Na + Cl₂ → NaCl
The plus sign indicates that two substances are involved in the reaction.
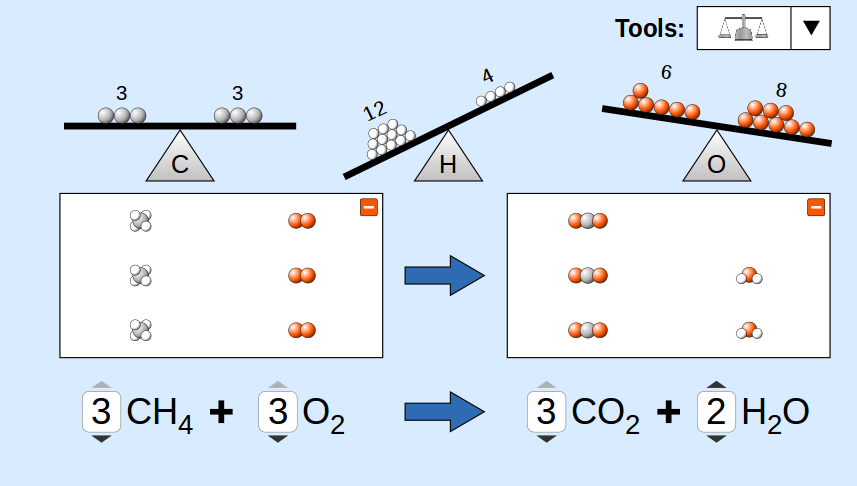
3. Balanced Chemical Equations




A balanced chemical equation has the same number of atoms of each element on both sides of the equation.
This requirement follows the law of conservation of mass, which states that matter cannot be created or destroyed during a chemical reaction.
For example:
Unbalanced equation:
H₂ + O₂ → H₂O
Balanced equation:
2H₂ + O₂ → 2H₂O
In the balanced equation:
Hydrogen atoms:
Left side = 4
Right side = 4
Oxygen atoms:
Left side = 2
Right side = 2
Balancing chemical equations is essential because it ensures that the equation accurately represents the conservation of atoms during a reaction.
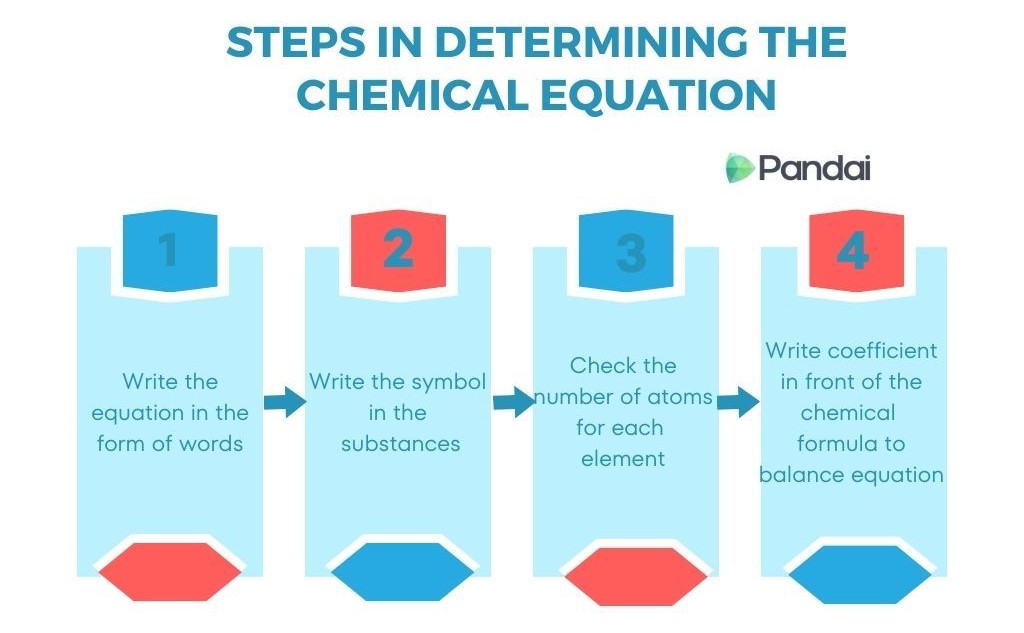
4. Steps for Balancing Chemical Equations



Balancing chemical equations involves adjusting coefficients so that the number of atoms of each element is equal on both sides of the equation.
Step 1: Write the skeletal equation.
Example:
Fe + O₂ → Fe₂O₃
Step 2: Count the atoms on each side.
Iron atoms:
Left = 1
Right = 2
Oxygen atoms:
Left = 2
Right = 3
Step 3: Adjust coefficients.
Balanced equation:
4Fe + 3O₂ → 2Fe₂O₃
Step 4: Verify atom balance.
Iron atoms:
Left = 4
Right = 4
Oxygen atoms:
Left = 6
Right = 6
This balanced equation now obeys the law of conservation of mass.
5. Types of Chemical Equations




Chemical equations represent different types of chemical reactions.
Synthesis Reactions
Two or more substances combine to form a single product.
Example:
2H₂ + O₂ → 2H₂O
Decomposition Reactions
A single compound breaks down into simpler substances.
Example:
2H₂O₂ → 2H₂O + O₂
Single Displacement Reactions
One element replaces another element in a compound.
Example:
Zn + CuSO₄ → ZnSO₄ + Cu
Double Displacement Reactions
Two compounds exchange ions to form new compounds.
Example:
AgNO₃ + NaCl → AgCl + NaNO₃
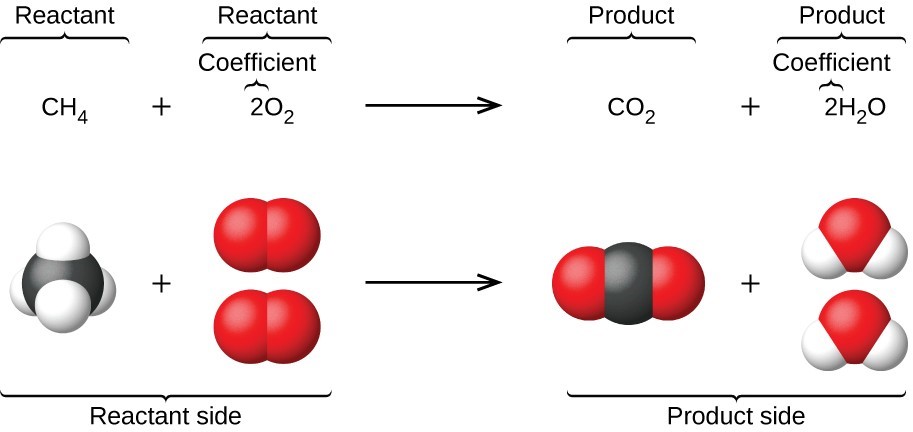
Combustion Reactions
A substance reacts with oxygen to produce energy, carbon dioxide, and water.
Example:
CH₄ + 2O₂ → CO₂ + 2H₂O
6. Physical States in Chemical Equations



Chemical equations often include symbols indicating the physical states of substances.
Common symbols include:
(s) → solid
(l) → liquid
(g) → gas
(aq) → aqueous solution
Example:
NaCl (aq) + AgNO₃ (aq) → AgCl (s) + NaNO₃ (aq)
These symbols provide additional information about the reaction environment.
7. Energy Changes in Chemical Equations




Chemical reactions involve energy changes.
Exothermic Reactions
Exothermic reactions release energy, usually in the form of heat.
Example:
Combustion reactions.
Endothermic Reactions
Endothermic reactions absorb energy from their surroundings.
Example:
Photosynthesis.
Energy changes may be included in chemical equations to indicate whether heat is released or absorbed.
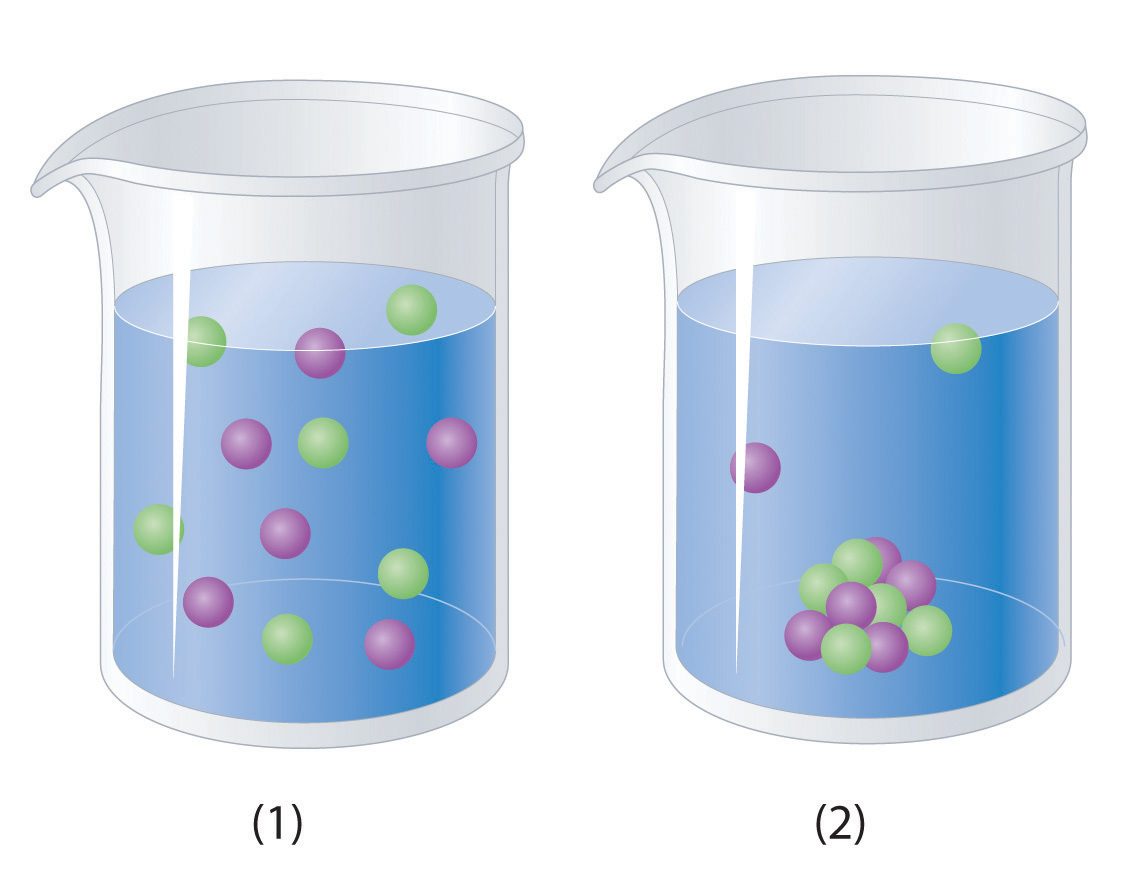
8. Molecular and Ionic Equations
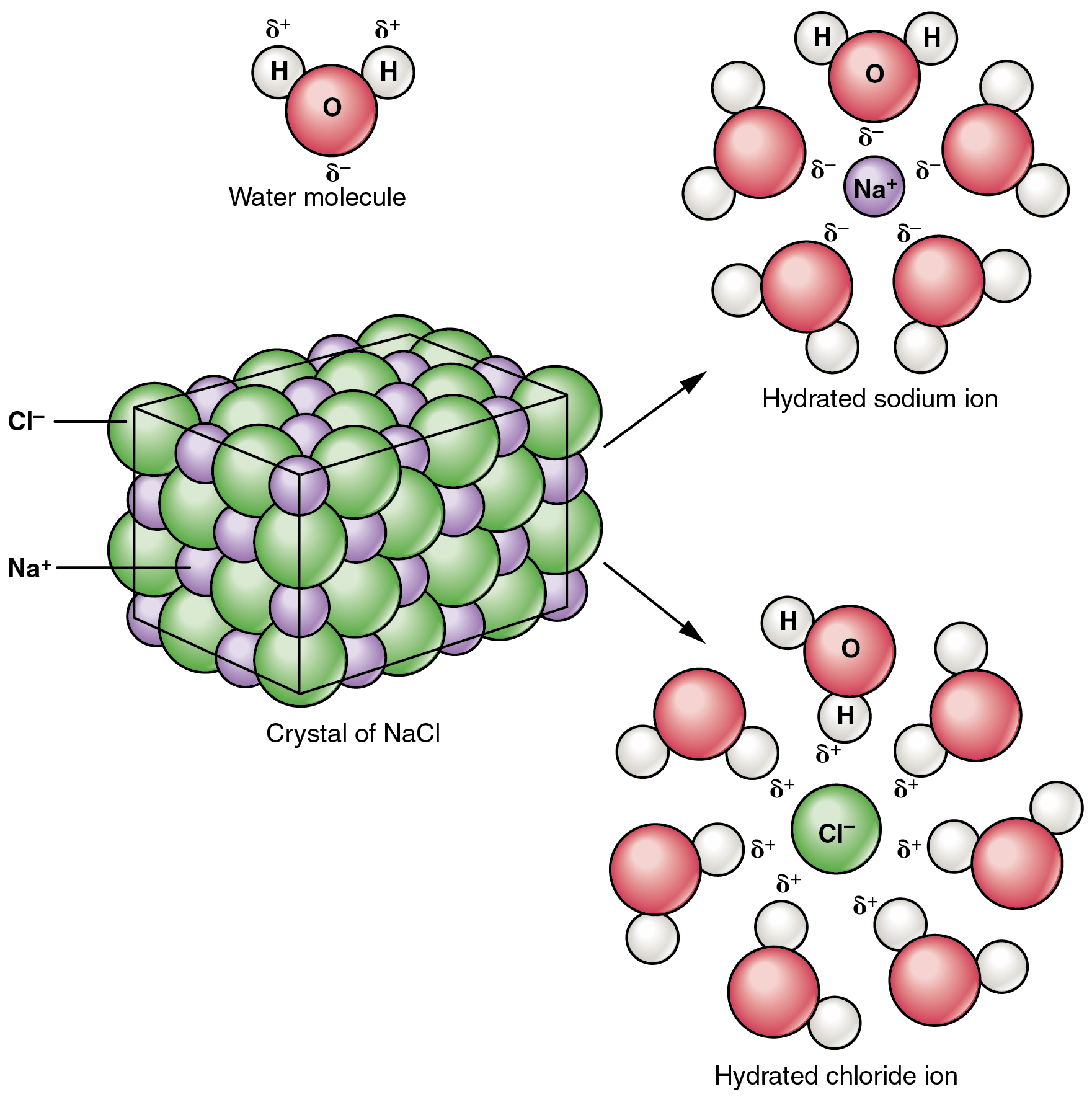



Chemical reactions in solutions can be represented using different forms of equations.
Molecular Equations
Show compounds as complete formulas.
Example:
AgNO₃ + NaCl → AgCl + NaNO₃
Ionic Equations
Show dissolved substances as ions.
Example:
Ag⁺ + NO₃⁻ + Na⁺ + Cl⁻ → AgCl + Na⁺ + NO₃⁻
Net Ionic Equations
Show only the ions involved in the reaction.
Example:
Ag⁺ + Cl⁻ → AgCl
These equations help simplify reactions occurring in aqueous solutions.
9. Importance of Chemical Equations
Chemical equations are fundamental tools in chemistry.
They help scientists:
- Understand chemical reactions
- Predict reaction outcomes
- Perform stoichiometric calculations
- Analyze reaction mechanisms
- Design industrial chemical processes
Chemical equations are also essential for communicating chemical information clearly.
10. Applications in Science and Industry
Chemical equations are widely used in many fields.
Industrial Chemistry
Chemical equations help design processes for manufacturing chemicals, fuels, and materials.
Environmental Science
They describe reactions involved in atmospheric chemistry and pollution.
Biochemistry
Biological processes such as respiration and photosynthesis are represented by chemical equations.
Medicine
Drug synthesis and metabolic pathways are explained using chemical reactions.
11. Limitations of Chemical Equations
Although chemical equations provide valuable information, they do not show all details of a reaction.
For example, they do not indicate:
- Reaction rates
- Intermediate steps
- Molecular orientations
- Reaction mechanisms
More advanced techniques such as kinetic studies and spectroscopy are required to study these aspects.
12. Conclusion
Chemical equations are symbolic representations that describe chemical reactions. They show the transformation of reactants into products and obey the law of conservation of mass.
By balancing chemical equations, chemists ensure that the number of atoms of each element remains constant during a reaction. Chemical equations also reveal important information about reaction types, physical states of substances, and energy changes.
Understanding chemical equations is essential for studying stoichiometry, predicting reaction outcomes, and designing chemical processes. From laboratory experiments to large-scale industrial production, chemical equations provide a universal language for describing chemical transformations.
Mastering chemical equations allows scientists to analyze and control chemical reactions with precision, making them a cornerstone of modern chemistry.