


Introduction
Band theory is a fundamental concept in solid-state physics that explains the behavior of electrons in solids and the electrical properties of materials such as conductors, semiconductors, and insulators. According to band theory, electrons in a solid do not occupy discrete atomic energy levels as they do in isolated atoms. Instead, the interaction between atoms in a crystal causes these energy levels to split and form continuous ranges of energy called energy bands.
Band theory helps explain why some materials conduct electricity easily, while others do not. It provides the theoretical foundation for understanding modern electronic devices such as transistors, diodes, and integrated circuits.
The development of band theory emerged from quantum mechanics and the study of electron behavior in periodic potentials within crystalline solids. Important contributions to this theory were made by scientists such as Felix Bloch and Arnold Sommerfeld.
Band theory is central to modern electronics, semiconductor physics, and materials science.
Energy Levels in Isolated Atoms
In isolated atoms, electrons occupy specific discrete energy levels determined by quantum mechanics. These levels correspond to electron orbitals around the nucleus.
Each energy level can hold a limited number of electrons according to quantum mechanical rules such as the Pauli exclusion principle.
When atoms are far apart, their electron energy levels remain independent.
However, when atoms come together to form a solid, the situation changes.
Formation of Energy Bands
When many atoms come close together in a crystal lattice, their electron orbitals overlap. This overlap causes the discrete energy levels of individual atoms to split into a very large number of closely spaced energy levels.
Because solids contain extremely large numbers of atoms (on the order of (10^{23})), these energy levels become so closely spaced that they form continuous energy ranges known as energy bands.
Between these bands, there may be energy ranges where no electron states exist. These are called band gaps.
Thus, the electronic structure of solids consists of:
- Valence band
- Conduction band
- Forbidden energy gap (band gap)
Valence Band
The valence band is the highest energy band that is normally occupied by electrons at absolute zero temperature.
Electrons in the valence band are responsible for chemical bonding between atoms in a crystal.
In many materials, electrons in the valence band are tightly bound to atoms and cannot move freely through the material.
Conduction Band
The conduction band is the energy band above the valence band.
Electrons in the conduction band are free to move throughout the crystal and contribute to electrical conduction.
When electrons gain enough energy, they can move from the valence band into the conduction band.
This transition allows electrons to move freely and carry electric current.
Forbidden Energy Gap (Band Gap)



The band gap is the energy difference between the valence band and the conduction band.
This gap determines whether a material behaves as a conductor, semiconductor, or insulator.
If the band gap is large, electrons cannot easily move to the conduction band.
If the band gap is small or nonexistent, electrons can move freely and conduct electricity.
Classification of Solids Using Band Theory
Band theory classifies materials into three major categories.
Conductors


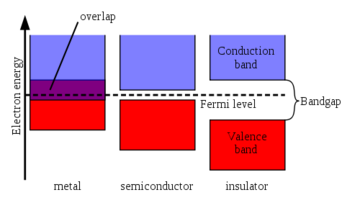
In conductors (such as metals), the valence band and conduction band overlap.
Because there is no energy gap, electrons can move freely between energy levels.
As a result:
- Electrical conductivity is high
- Electrons move easily through the material
Examples of conductors include:
- Copper
- Aluminum
- Silver
Insulators


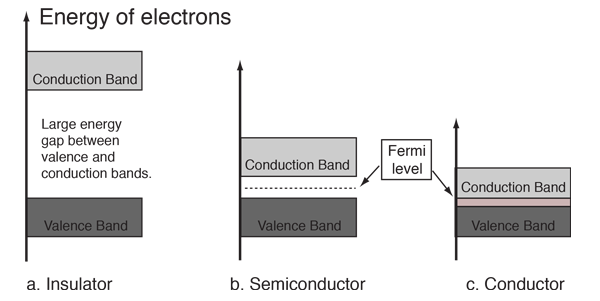
In insulators, the band gap between the valence band and conduction band is very large.
Because of this large gap, electrons cannot easily move to the conduction band.
Consequently:
- Electrical conductivity is extremely low.
Examples of insulators include:
- Glass
- Rubber
- Diamond
Semiconductors



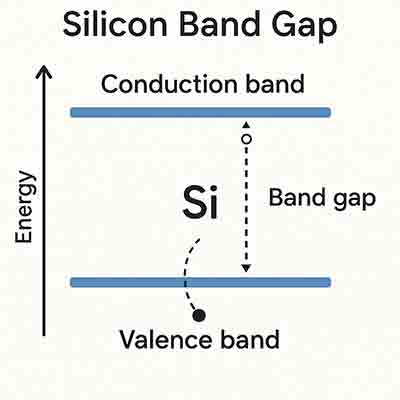
Semiconductors have a small band gap between the valence and conduction bands.
At low temperatures, they behave like insulators.
However, when energy is supplied (such as heat or light), electrons can move into the conduction band.
This allows semiconductors to conduct electricity under certain conditions.
Examples include:
- Silicon
- Germanium
- Gallium arsenide
Semiconductors are essential for modern electronic devices.
Electron and Hole Concept
When an electron moves from the valence band to the conduction band, it leaves behind an empty state called a hole.
A hole behaves like a positively charged particle.
Electrical conduction in semiconductors occurs through the movement of:
- Electrons in the conduction band
- Holes in the valence band
Intrinsic Semiconductors
An intrinsic semiconductor is a pure semiconductor without impurities.
In intrinsic semiconductors:
- Electron-hole pairs are generated by thermal energy.
Examples include pure silicon crystals.
The number of electrons equals the number of holes.
Extrinsic Semiconductors



Extrinsic semiconductors are created by adding impurities to intrinsic semiconductors.
This process is called doping.
Two types exist:
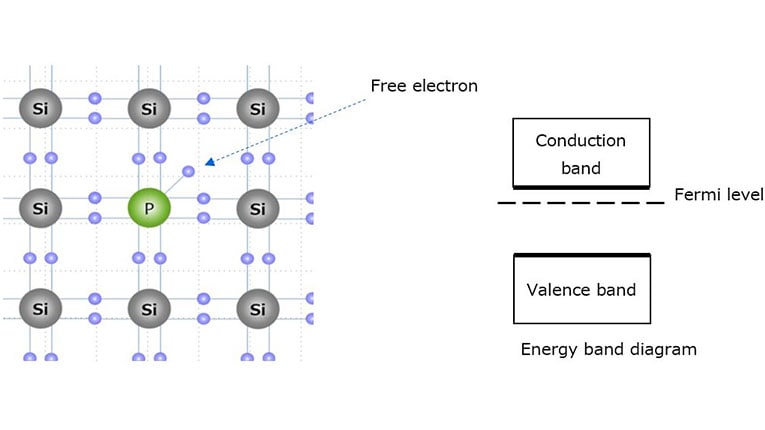
n-Type Semiconductor
Doping with atoms that provide extra electrons.
Example: phosphorus in silicon.
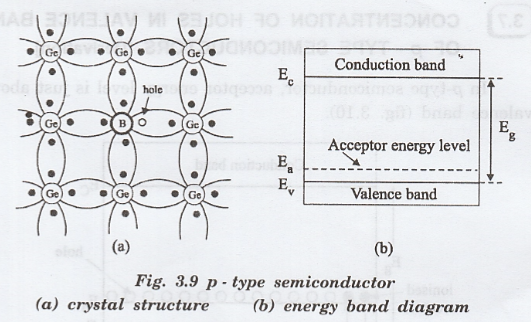
p-Type Semiconductor
Doping with atoms that create holes.
Example: boron in silicon.
Doping increases electrical conductivity.
Fermi Energy Level
The Fermi level represents the highest occupied energy level at absolute zero temperature.
In conductors:
- The Fermi level lies within an energy band.
In semiconductors:
- The Fermi level lies within the band gap.
The position of the Fermi level determines electrical behavior.
Band Theory and Temperature
Temperature strongly affects electron behavior in solids.
As temperature increases:
- More electrons gain energy.
- More electrons move into the conduction band.
This increases electrical conductivity in semiconductors.
In metals, however, conductivity slightly decreases with temperature due to increased atomic vibrations.
Band Structure and Optical Properties
Band theory also explains optical properties of materials.
When photons interact with solids, electrons may absorb energy and move across the band gap.
This process determines:
- Color
- Transparency
- Optical absorption
For example:
- Large band gaps → transparent materials
- Small band gaps → colored materials.
Applications of Band Theory
Band theory is fundamental for many modern technologies.
Semiconductor Devices
Electronic devices such as:
- Diodes
- Transistors
- Integrated circuits
operate based on band theory.
Solar Cells
Solar cells convert light into electricity through electron excitation across band gaps.
LEDs
Light-emitting diodes produce light when electrons recombine with holes in semiconductors.
Lasers
Semiconductor lasers rely on electron transitions between energy bands.
Band Theory in Modern Physics
Band theory continues to evolve with modern research.
Scientists study complex band structures in advanced materials such as:
- Graphene
- Topological insulators
- Superconductors
These materials exhibit unusual electronic properties that expand the understanding of solid-state physics.
Experimental Techniques
Scientists study band structures using advanced techniques such as:
- Photoelectron spectroscopy
- X-ray diffraction
- Electron microscopy
These methods allow precise measurement of electronic structures in solids.
Conclusion
Band theory provides a powerful framework for understanding the electronic properties of solids. It explains how energy levels in individual atoms combine to form continuous energy bands in crystalline materials. The presence or absence of a band gap determines whether a material behaves as a conductor, semiconductor, or insulator.
This theory is fundamental to modern electronics and materials science. It explains the operation of semiconductors, the behavior of metals, and the insulating properties of certain materials. Advances in band theory have led to the development of numerous technologies, including computers, communication devices, and renewable energy systems.
As research continues, band theory remains a central concept in solid-state physics and plays a key role in the design of new materials with advanced electronic and optical properties.
