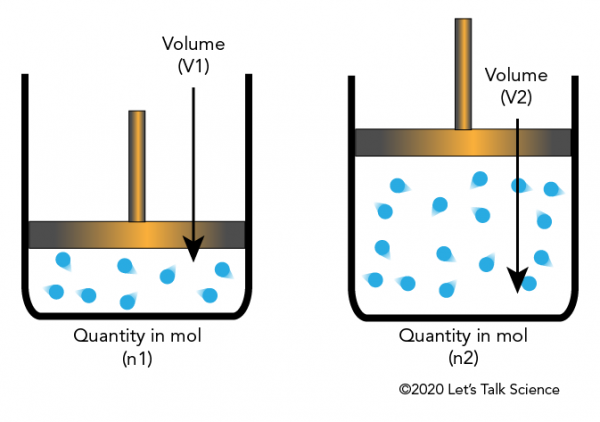
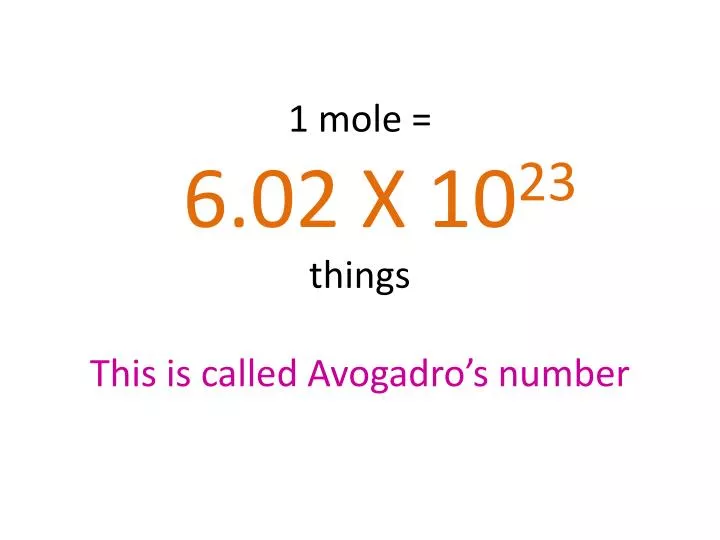1. Introduction to Stoichiometry
Stoichiometry is a branch of chemistry that deals with the quantitative relationships between reactants and products in chemical reactions. It allows chemists to calculate the exact amounts of substances consumed and produced during chemical reactions.
The term stoichiometry comes from the Greek words stoicheion meaning element and metron meaning measure. Essentially, stoichiometry is the science of measuring elements and compounds involved in chemical reactions.
When a chemical reaction occurs, atoms are rearranged to form new substances. According to the law of conservation of mass, atoms cannot be created or destroyed during a chemical reaction. Instead, they are redistributed among the reactants and products. Stoichiometry provides the mathematical framework for understanding these relationships.
Stoichiometric calculations allow scientists to determine:
- How much reactant is required for a reaction
- How much product will be produced
- Which reactant will be consumed first
- The theoretical yield of a reaction
- The efficiency of chemical reactions
Stoichiometry is fundamental to chemistry because it connects chemical equations with measurable quantities of substances. Without stoichiometry, it would be impossible to accurately design chemical reactions in laboratories, industries, or pharmaceutical production.
2. Balanced Chemical Equations


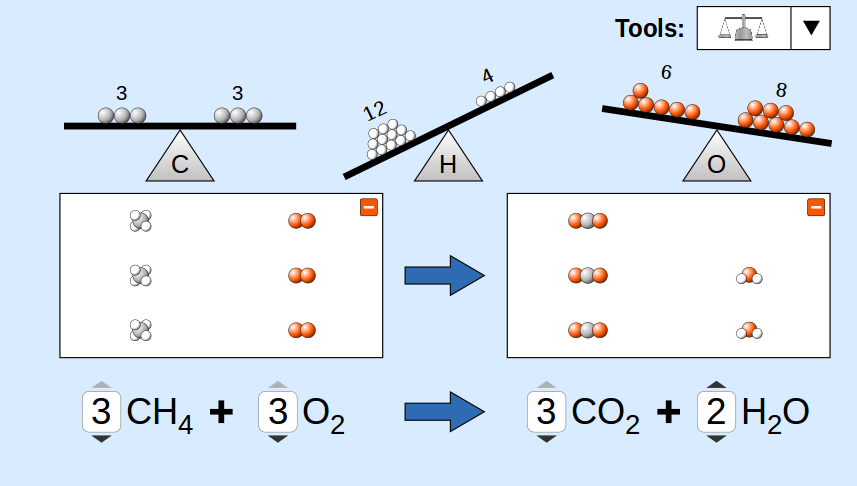
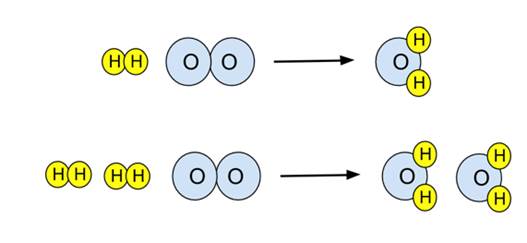
Stoichiometry begins with a balanced chemical equation. A chemical equation represents a chemical reaction by showing the reactants and products involved.
For example:
2H₂ + O₂ → 2H₂O
In this equation:
- Hydrogen and oxygen are reactants.
- Water is the product.
Balancing a chemical equation ensures that the number of atoms of each element is the same on both sides of the equation. This reflects the law of conservation of mass.
In the above reaction:
Hydrogen atoms:
Left side = 4
Right side = 4
Oxygen atoms:
Left side = 2
Right side = 2
Balanced equations provide the mole ratios used in stoichiometric calculations.
3. Mole Ratios in Stoichiometry

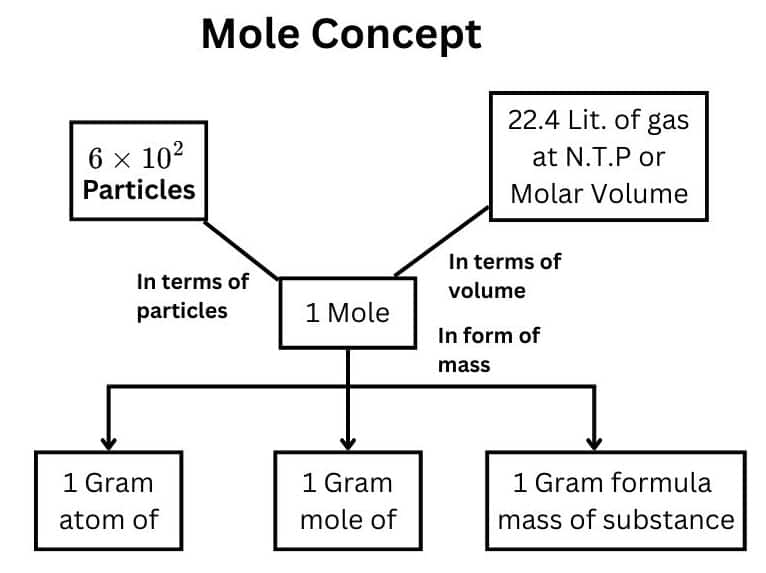


Mole ratios are the relationships between the quantities of substances in a balanced chemical equation.
From the equation:
2H₂ + O₂ → 2H₂O
We can determine the following mole ratios:
2 moles H₂ react with 1 mole O₂
2 moles H₂ produce 2 moles H₂O
1 mole O₂ produces 2 moles H₂O
These ratios allow chemists to convert between quantities of reactants and products.
For example, if 4 moles of hydrogen react with oxygen, we can determine the amount of water produced using the mole ratio.
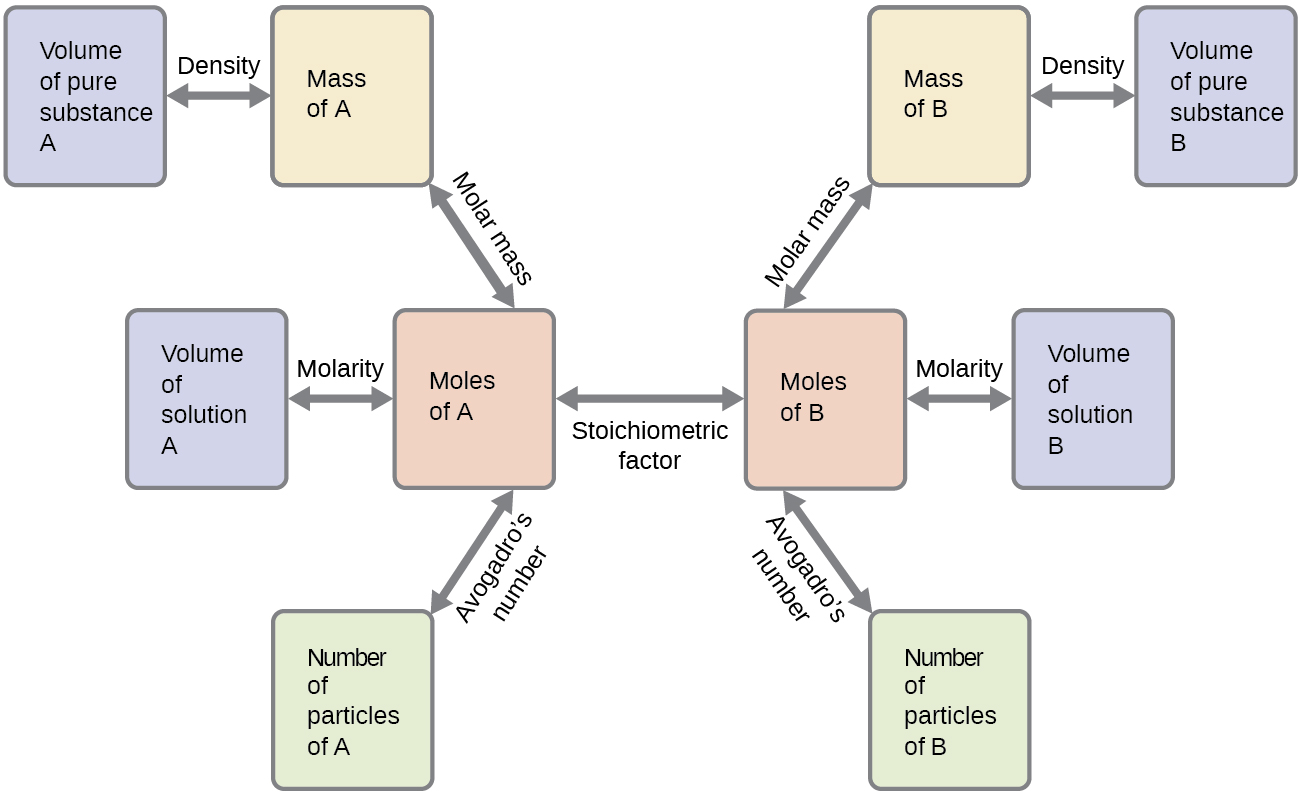
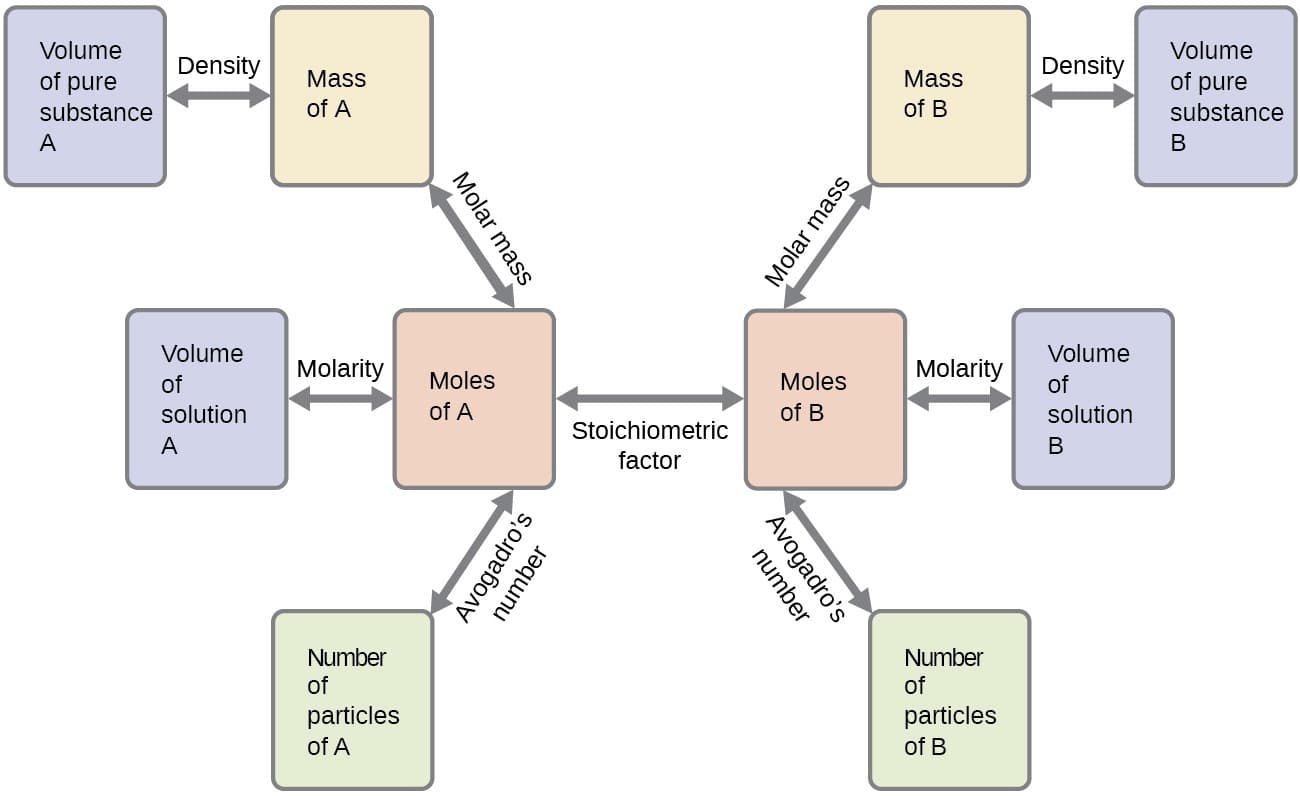
Stoichiometric calculations often involve converting between:
- Moles of reactants
- Moles of products
These conversions form the basis of many chemical calculations.
4. Stoichiometric Calculations
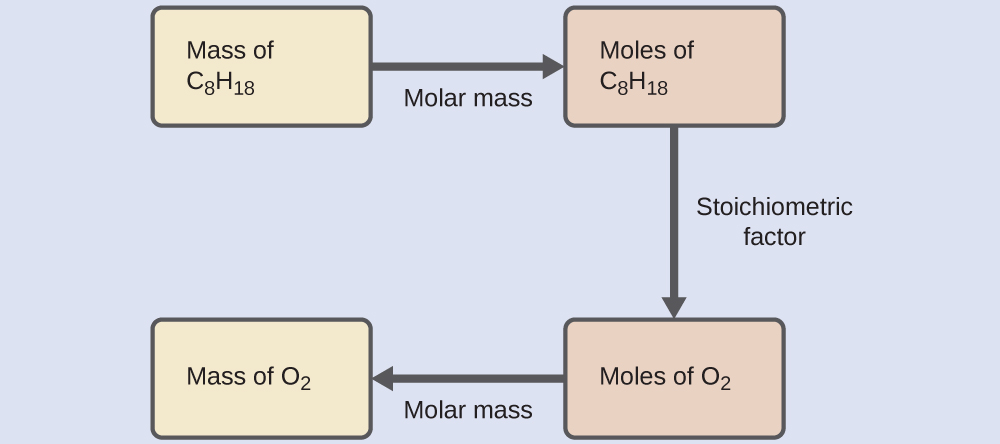

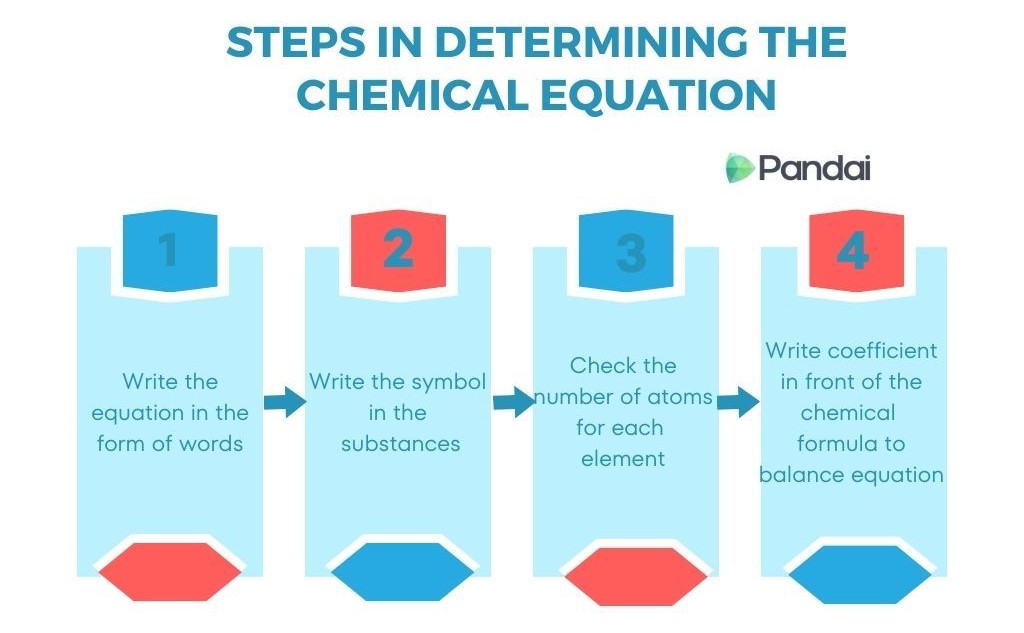

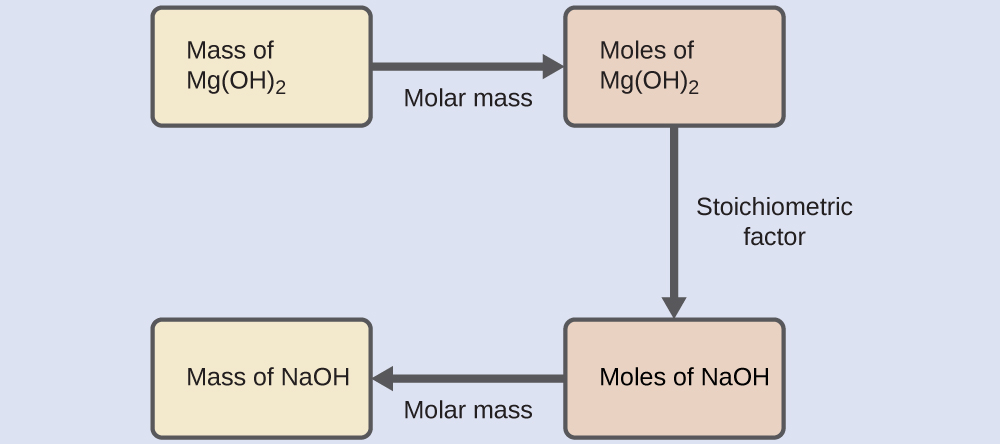
Stoichiometric calculations typically follow several steps.
Step 1: Write a balanced chemical equation.
Example:
2H₂ + O₂ → 2H₂O
Step 2: Convert known quantities to moles.
Example:
Mass ÷ molar mass = moles
Step 3: Use mole ratios from the balanced equation.
Convert moles of one substance to moles of another.
Step 4: Convert moles to desired units.
Possible units include:
- Mass
- Volume
- Number of particles
These steps allow chemists to calculate quantities involved in chemical reactions.
5. Limiting and Excess Reactants
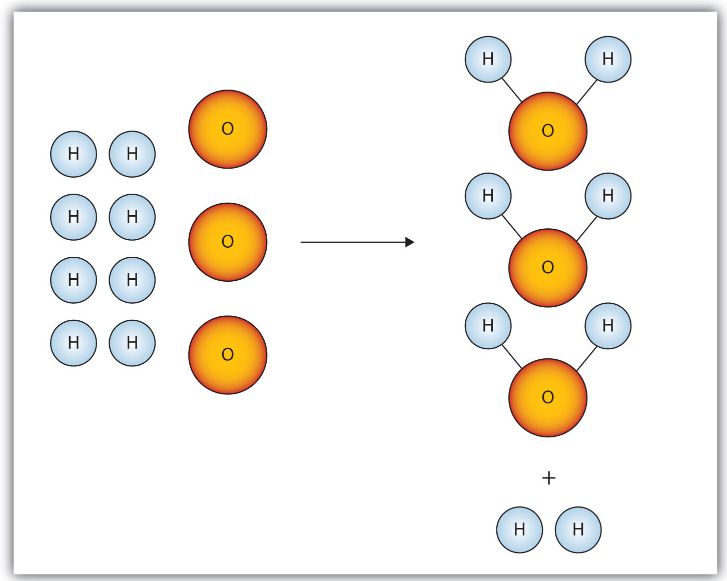



In many reactions, reactants are not present in exact stoichiometric proportions.
One reactant is consumed completely, limiting the amount of product that can form. This reactant is called the limiting reactant.
The other reactant remains after the reaction is complete and is called the excess reactant.
Example:
If hydrogen and oxygen react to form water, the reactant that runs out first determines how much water can form.
Identifying the limiting reactant is important in chemical manufacturing because it determines the maximum possible product yield.
6. Theoretical Yield
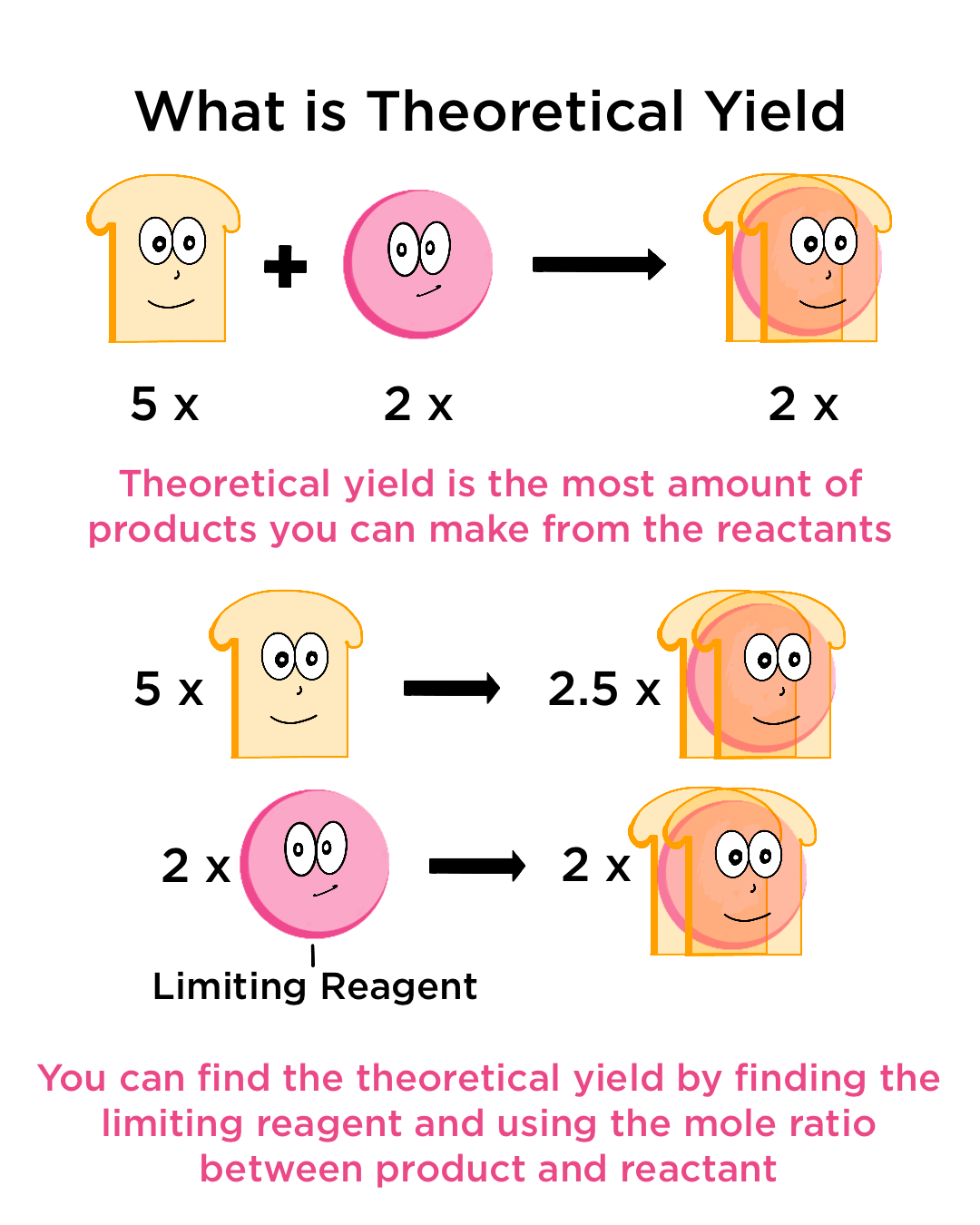
The theoretical yield is the maximum amount of product that can be formed from a given amount of reactants according to stoichiometric calculations.
It assumes that:
- The reaction proceeds perfectly
- No side reactions occur
- All reactants are converted to products
Theoretical yield is calculated using mole ratios from the balanced equation.
For example:
From the reaction:
2H₂ + O₂ → 2H₂O
If 2 moles of hydrogen react with oxygen, the theoretical yield of water is 2 moles.
7. Actual Yield and Percent Yield



In real chemical reactions, the amount of product formed is usually less than the theoretical yield.
The amount actually obtained in an experiment is called the actual yield.
The efficiency of a reaction is measured using percent yield.
Formula:
Percent yield = (Actual yield ÷ Theoretical yield) × 100
Example:
If theoretical yield = 10 g
Actual yield = 8 g
Percent yield = 80%
Percent yield indicates how efficient a chemical reaction is.
8. Stoichiometry with Gases




Stoichiometric calculations can also involve gases.
At standard temperature and pressure (STP):
1 mole of gas occupies 22.4 liters.
This relationship allows chemists to calculate gas volumes in reactions.
Example:
If a reaction produces 2 moles of oxygen gas, the volume at STP would be:
2 × 22.4 L = 44.8 L
Gas stoichiometry is important in industrial processes and environmental chemistry.
9. Stoichiometry in Solutions




Stoichiometry is also applied to reactions occurring in solutions.
In such cases, concentrations are often expressed using molarity.
Molarity = moles of solute ÷ liters of solution
Stoichiometry with solutions is commonly used in titration experiments, where the concentration of an unknown solution is determined using a reaction with a known solution.
For example, acid–base titrations allow chemists to determine the concentration of acids or bases.
10. Applications of Stoichiometry
Stoichiometry has many important applications in science and industry.
Chemical Manufacturing
Industries rely on stoichiometric calculations to determine the correct amounts of reactants needed to produce chemicals efficiently.
Pharmaceutical Production
Precise stoichiometric ratios are necessary to produce medicines with correct chemical composition.
Environmental Chemistry
Stoichiometry helps analyze pollutant concentrations and chemical reactions in the environment.
Agriculture
Fertilizer production and soil chemistry rely on stoichiometric calculations.
Food Chemistry
Stoichiometry helps understand chemical changes during cooking and food processing.
11. Importance of Stoichiometry
Stoichiometry is one of the most essential tools in chemistry because it connects chemical equations with measurable quantities.
It allows chemists to:
- Predict reaction outcomes
- Determine reaction efficiency
- Design industrial chemical processes
- Analyze laboratory experiments
- Understand environmental chemical processes
Without stoichiometry, quantitative chemical analysis would not be possible.
12. Conclusion
Stoichiometry is the quantitative foundation of chemical reactions. By using balanced chemical equations and mole ratios, chemists can calculate the amounts of reactants required and the products formed during reactions.
The mole concept, molar mass, and Avogadro’s number are essential tools used in stoichiometric calculations. These concepts allow scientists to convert between mass, moles, and number of particles.
Stoichiometry also helps identify limiting reactants, determine theoretical and actual yields, and calculate percent yield to evaluate reaction efficiency.
From laboratory experiments to large-scale industrial processes, stoichiometry plays a crucial role in ensuring accurate chemical measurements and efficient reactions. Understanding stoichiometry is therefore essential for mastering chemistry and applying it to real-world problems.